Medical Information (from PDR) about the components
of
KALA Health MSM, Glucosamine and
Chondroitin Joint Support Combo
Methylsulfonylmethane (MSM)
Glucosamine
Chondroitin Sulfate
Vitamin C
Manganese
Molybdenum
Methylsulfonylmethane (MSM)
DESCRIPTION
Methylsulfonylmethane, abbreviated MSM, is an organic sulfur-containing compound that occurs naturally in a variety of fruits, vegetables, grains and in animals, including humans in at least trace amounts. MSM has also been found in such plants as Equisetem arvense, also known as horsetail. The biological role of MSM, if any, is not known. MSM is a metabolite of dimethyl sulfoxide or DMSO (see Dimethyl Sulfoxide). It is believed that some of the possible effects of DMSO could be attributed to MSM.
MSM is a water-soluble, solid compound. It is also known as dimethyl sulfone, DMSO2, sulfonylbismethane and methyl sulfone.
ACTIONS AND PHARMACOLOGY
ACTIONS
None known.
PHARMACOKINETICS
Little is known about the pharmacokinetics of MSM in humans. Sulfur from MSM was found to be incorporated into protein methionine and cysteine when fed to guinea pigs. MSM was also detected in the brain of a normal 62-year old male, following its ingestion, using in vivo proton magnetic resonance spectroscopy. Thus, it appears that MSM gets absorbed and can cross the blood-brain barrier.
INDICATIONS AND USAGE
Claims for MSM include pain relief, particularly in arthritis, immune modulation in autoimmune disorders, muscle repair, sleep aid and diabetes therapy. There is preliminary research suggesting some possible MSM anti-cancer effects.
RESEARCH SUMMARY
Two animal studies showed that MSM and other bipolar solvents can prolong latency period to time of tumor appearance in chemically induced animal model cancers. In one of these studies, there was no effect on tumor incidence; in the other, MSM seemed to reduce the incidence of poorly differentiated tumors. More research is indicated.
CONTRAINDICATIONS, PRECAUTIONS ADVERSE REACTIONS
CONTRAINDICATIONS
None known.
PRECAUTIONS
MSM should be avoided by pregnant women and nursing mothers.
ADVERSE REACTIONS
Reported adverse reactions include nausea, diarrhea and headache.
OVERDOSAGE
There are no reports of overdosage.
DOSAGE AND ADMINISTRATION
Doses used are typically 1 to 8 grams daily.
HOW SUPPLIED
Powder — 2600 mg/0.5 teaspoonful
Tablets — 1000 mg
LITERATURE
Childs SJ. Dimethyl sulfone (DMSO2) in the treatment of interstitial cystitis. Urol Clin North Am. 1994; 21:85-98.
Kandorf H, Chirra AR, De Gruccio A, Girman DJ. Dimethyl sulfoxide modulation of diabetes onset in NOD mice. Diabetes. 1989; 38:194-197.
Kocsis JJ, Harkaway S, Snyder R. Biological effects of the metabolites of dimethyl sulfoxide. Ann NY Acad Sci. 1975; 243:104-109.
Layman DL. Growth inhibitory effects of dimethyl sulfoxide and dimethyl sulfone on vascular smooth muscle and endothelial cells in vitro. In Vitro Cell Dev Biol. 1987; 23:422-428.
Morton JI, Siegel BV. Effects of oral dimethyl sulfoxide and dimethyl sulfone on murine autoimmune lymphoproliferative disease. Proc Soc Exp Biol Med. 1986; 183; 227-230.
O’Dwyer PJ, McCabe DP, Sickle-Santanello BJ, et al. Use of polar solvents in chemoprevention of 1, 2-dimethylhydrazine-induced colon cancer. Cancer. 1988; 62:944-948.
Pearson TW, Dawson HJ, Lackey HB. Natural occurring levels of dimethyl sulfoxide in selected fruits, vegetables, grains and beverages. J Agric Food Chem. 1989; 29:1089-1091.
Richmond VL, Incorporation of methylsulfonylmethane sulfur into guinea pig serum proteins. Life Sci. 1986; 39:263-268.
Rose SE, Chalk JB, Galloway GJ, Doddrell DM. Detection of dimethyl sulfone in the human brain by in vivo proton magnetic resonance spectroscopy. Magn Reson Imaging. 2000; 18:95-98.
Glucosamine
TRADE NAMES
Glucosamine is available from numerous manufacturers generically. Branded products include Aflexa (McNeil Consumer), Natures Blend Glucosamine (National Vitamin Co.), GS-500 (Enzymatic Therapy), Glucosamine Complex (Schiff), Maxi GS (Maxi-Health Research), NAG (Twinlab).
DESCRIPTION
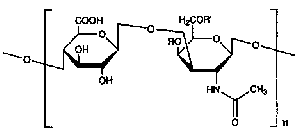
Glucosamine is an amino monosaccharide found in chitin, glycoproteins and glycosaminoglycans (formerly known as mucopolysaccharides) such as hyaluronic acid and heparan sulfate. Glucosamine is also known as 2-amino-2-deoxyglucose, 2-amino-2-deoxy-beta-D-glucopyranose and chitosamine. Glucosamine has the following chemical structure:
Glucosamine
Glucosamine is available commercially as a nutritional supplement in three forms: glucosamine hydrochloride or glucosamine HCl, glucosamine sulfate and N-acetyl-glucosamine.
At neutral as well as physiologic pH, the amino group in glucosamine is protonated, resulting in its having a positive charge. Salt forms of glucosamine contain negative anions to neutralize the charge. In the case of glucosamine hydrochloride, the anion is chloride, and in glucosamine sulfate the anion is sulfate. N-acetylglucosamine is a delivery form of glucosamine in which the amino group is acetylated, thus neutralizing its charge. To date, most of the clinical studies examining the effect of glucosamine on osteoarthritis have been performed with either the sulfate or the chloride salts of glucosamine. All three forms are water soluble.
The glucosamine used in supplements is typically derived from marine exoskeletons. Synthetic glucosamine is also available.
ACTIONS AND PHARMACOLOGY
ACTIONS
The actions of supplemental glucosamine have yet to be clarified. It may play a role in the promotion and maintenance of the structure and function of cartilage in the joints of the body. Glucosamine may also have anti-inflammatory properties.
MECHANISM OF ACTION
Until the specific actions of supplemental glucosamine are determined, the mechanism of action in relieving arthritic pain and in repair of cartilage is a matter of speculation. However, we do know a great deal about the biochemistry of the molecules in which glucosamine is found. Biochemically, glucosamine is involved in glycoprotein metabolism. Glycoproteins, known as proteoglycans, form the ground substance in the extra-cellular matrix of connective tissue. Proteoglycans are polyanionic substances of high-molecular weight and contain many different types of heteropolysaccharide side-chains covalently linked to a polypeptide-chain backbone. These polysaccharides make up to 95% of the proteoglycan structure. In fact, chemically, proteoglycans resemble polysaccharides more than they do proteins.
The polysaccharide groups in proteoglycans are called glycosaminoglycans or GAGs. GAGs include hyaluronic acid, chondroitin sulfate, dermatan sulfate, keratan sulfate, heparin and heparan sulfate. All of the GAGs contain derivatives of glucosamine or galactosamine.
Glucosamine derivatives are found in hyaluronic acid, keratan sulfate and heparan sulfate. Chondroitin sulfate contains derivatives of galactosamine.
The glucosamine-containing glycosaminoglycan hyaluronic acid is vital for the function of articular cartilage. GAG chains are fundamental components of aggrecan found in articular cartilage. Aggrecan confers upon articular cartilage shock-absorbing properties. It does this by providing cartilage with a swelling pressure that is restrained by the tensile forces of collagen fibers. This balance confers upon articular cartilage the deformable resilience vital to its function.
In the early stages of degenerative joint disease, aggrecan biosynthesis is increased. However, in later stages, aggrecan synthesis is decreased, leading eventually to the loss of cartilage resiliency and to most of the symptoms that accompany osteoarthritis.
During the progression of osteoarthritis, exogenous glucosamine may have a beneficial role. It is known that, in vitro, chondrocytes do synthesize more aggregan when the culture medium is supplemented with glucosamine. N-acetylglucosamine is found to be less effective in these in vitro studies. Glucosamine has also been found to have antioxidant activity and to be beneficial in animal models of experimental arthritis.
The counter anion of the glucosamine salt (i.e. chloride or sulfate) is unlikely to play any role in the action or pharmacokinetics of glucosamine. Further, the sulfate in glucosamine sulfate supplements should not be confused with the glucosamine sulfate found in such GAGs as keratan sulfate and heparan sulfate. In the case of the supplement, sulfate is the anion of the salt. In the case of the above GAGs, sulfate is present as an ester. Also, there is no glucosamine sulfate in chondroitin sulfate.
PHARMACOKINETICS
Pharmacokinetics of glucosamine are derived primarily from animal studies. About 90% of glucosamine administered orally as a glucosamine salt gets absorbed from the small intestine, and from there it is transported via the portal circulation to the liver. It appears that a significant fraction of the ingested glucosamine is catabolized by first-pass metabolism in the liver. Free glucosamine is not detected in the serum after oral intake, and it is not presently known how much of an ingested dose is taken up in the joints in humans. Some uptake in the articular cartilage is seen in animal studies.
INDICATIONS
Glucosamine may be indicated for the treatment and prevention of osteoarthritis, either by itself or in combination with chondroitin sulfate (see Chondroitin Sulfate).
RESEARCH SUMMARY
Two recent meta-analyses have confirmed that glucosamine is useful in the treatment of osteoarthritis. One of these meta-analyses included all double-blind, placebo-controlled trials that lasted four weeks or longer. This meta-analysis also included trials that studied the effects of chondroitin sulfate (see Chondroitin Sulfate). In all, there were l3 of these studies (six involving glucosamine and seven involving chondroitin sulfate).
All l3 studies found positive results in hip or knee osteoarthritis. The authors of the meta-analysis judged a trial positive if there was 25% or more improvement in the treatment group compared with placebo. The Levesque Index and global pain scores were used to assess improvement. Very significant improvement was associated with both glucosamine (39.5%) and chondroitin sulfate (40.2%), compared with placebo.
In another recent meta-analysis of nine randomized, controlled trials of glucosamine, glucosamine was significantly superior to placebo in seven of the studies and was superior to ibuprofen and equal to ibuprofen in the other two studies.
Recently, a long-term, randomized placebo-controlled trial of glucosamine sulfate’s effects on osteoarthritis ended with the conclusion that the supplement halts progression of structural joint damage and reduces symptoms of those with osteoarthritis of the knee. The study involved 212 patients 50 years or older who received 1500 milligrams of glucosamine sulfate daily or placebo.
Radiographic evidence, at a three-year followup, showed joint space narrowing–the prime indicator of arthritic joint damage–in the placebo group consistent with what has been documented to be typical in untreated osteoarthritis. The glucosamine-supplemented subjects, on the other hand, showed only a non-significant increase in joint space at the same three-year followup.
There has been one study demonstrating an apparent synergistic effect using glucosamine and chondroitin together. The combination was more effective than either substance alone in inhibiting progression of degenerative cartilage lesions in an experimental study.
Clinical research is needed to determine if this effect is truly synergistic, additive or non-existent. The National Institutes of Health has started a large, multi-center study that may shed further light on this issue.
It is probably not surprising that glucosamine may be helpful in osteoarthritis. Glucosamine is crucial for the construction of glycosaminoglycans (GAGs) in articular cartilage. Reduced GAG content in osteoarthritic cartilage matrix corresponds with the severity of osteoarthritis. Oral glucosamine appears to be capable of prompting the chondrocytes to secrete more GAGs. This knowledge, derived from animal and in vitro studies, has prompted clinical trials of glucosamine in osteoarthritis.
CONTRAINDICATIONS, PRECAUTIONS, ADVERSE REACTIONS
CONTRAINDICATIONS
There are no known contraindications to glucosamine supplementation.
WARNINGS AND PRECAUTIONS
Glucosamine may increase insulin resistance. Glucosamine increases insulin resistance in normal and experimentally diabetic animals. In these animals, intravenous glucosamine significantly decreases the rate of glucose uptake in skeletal muscle. In animals given oral glucosamine, this is not observed.
Those with type 2 diabetes and those who are overweight and have problems with glucose tolerance should have their blood sugars carefully monitored if they use glucosamine supplements. Because of insufficient safety data, children, pregnant women and nursing mothers should avoid using glucosamine.
ADVERSE REACTIONS
Side effects that have been reported are mainly mild gastrointestinal complaints such as heartburn, epigastric distress and diarrhea. No allergic reactions have been reported including sulfa-allergic reactions to glucosamine sulfate.
INTERACTIONS
Glucosamine may increase insulin resistance and consequently affect glucose tolerance. Diabetics who, under medical advisement, decide to use glucosamine supplements will need to monitor their blood glucose and may need to adjust the doses of the medications they take to control blood glucose. This needs to be done under medical supervision. No other drug, nutritional supplement, food or herb interaction is known.
OVERDOSAGE
None known.
DOSAGE AND ADMINISTRATION
The three forms of glucosamine available commercially are glucosamine hydrochloride, glucosamine sulfate and N-acetyl glucosamine. The usual dose used by those with osteoarthritis is l,500 milligrams daily in divided doses. These three forms of glucosamine are available in 500 milligram capsules.
The amount of glucosamine base varies with the supplemental form. Pure glucosamine hydrochloride is about 83% in glucosamine base, pure glucosamine sulfate is about 65% in glucosamine base, and pure N-acetyl glucosamine, about 75% in glucosamine base. It is important that all clinical studies standardize the glucosamine dose of the form used to glucosamine base.
Supplements are available containing glucosamine and low-molecular-weight chondroitin sulfate. (See Chondroitin Sulfate.)
It usually takes several weeks of supplementation before effects, if any, are noted.
HOW SUPPLIED
Capsules — 500 mg, 550 mg, 750 mg, 1000 mg
Powder
Liquid — 500 mg/5 mL
Tablets — 340 mg, 500 mg, 1000 mg
LITERATURE
Deal CL, Moskowitz RW. Nutraceuticals as therapeutic agents in osteoarthritis. The role of glucosamine, chondroitin sulfate, and collagen hydrolysate. Rheum Dis Clin North Am. 1999; 25:379-395.
Drovanti A, Bignamini AA, Rovati AL. Therapeutic activity of oral glucosamine sulfate in osteoarthritis, a placebo-controlled double-blind investigation. Clin Ther. 1980; 3:260-272.
Houpt JB, McMillan R, Wein C, Paget-Dello SD. Effect of glucosamine hydrochloride in the treatment of pain of osteoarthritis of the knee. J Rheumatol. 1999; 26:2423-2430.
Leffler CT, Philippi AF, Leffler SG, et al. Glucosamine, chondroitin, and manganese ascorbate for degenerative joint disease of the knee or low back: a randomized double-blind, placebo-controlled pilot study. Mil Med. 1999; 64:85-91.
McClain DA, Crook, ED. Hexosamines and insulin resistance. Diabetes. 1996; 45:l003-l006.
Noack W, Fischer, M., Forster, KK, et al. Glucosamine sulfate in osteoarthritis of the knee. Osteoarthritis Cartilage. 1994; 2:51-59.
Pujalte JM, Llavore EP, Ylescupidez FR. Double-blind evaluation of oral glucosamine sulfate in the basic treatment of osteoarthritis. Curr Med Res Opin. 1980; 7:110-114.
Reichelt A, Forster K, Fisher M, et al. Efficacy and safety of intramuscular glucosamine sulfate in osteoarthritis of the knee. A randomized, placebo-controlled, double-blind study. Arzneimittelforschung. 1999; 44:75-80.
Setnikar I, Giacchetti C, Zanolo G. Pharmacokinetics of glucosamine in the dog and in man. Arzneimittelforschung. 1986; 36:729-735.
Setnikar I, Palumbo R, Canali S, Zanolo G. Pharmacokinetics of glucosamine in man. Arzneimittelforschung.1993; 43:1109-1113.
Towheed TE, Anastassiades TP. Glucosamine and chondroitin for treating symptoms of osteoarthritis. Evidence is widely touted but incomplete. JAMA. 2000; 283:1483-1484.
Towheed TE, Anastassiades TP. Glucosamine therapy for osteoarthritis. Editorial. J Rheumatol l999; 26:2294-2297.
Chondroitin Sulfate
TRADE NAMES
Chondroitin sulfate is available from numerous manufacturers generically. Branded products include Ramott (Key Company) Chondroitin Sulfate Support (Natural Treasures), CSA (Twinlab), Chonflex (American Health).
DESCRIPTION
Chondroitin sulfate belongs to a family of heteropolysaccharides called glycosaminoglycans or GAGs. Glycosaminoglycans were formerly known as mucopolysaccharides. GAGs in the form of proteoglycans comprise the ground substance in the extracellular matrix of connective tissue. Chondroitin sulfate is made up of linear repeating units containing D-galactosamine and D-glucuronic acid. Chondroitin sulfate is found in humans in cartilage, bone, cornea, skin and the arterial wall. This type of chondroitin sulfate is sometimes referred to as chondroitin sulfate A or galactosaminoglucuronoglycan sulfate. The amino group of galactosamines in the basic unit of chondroitin sulfate A is acetylated, yielding N-acetyl-galactosamine; there is a sulfate group esterified to the 4-position in N-acetyl-galactosamine. (Chondroitin sulfate A is also sometimes called chondroitin 4-sulfate.) The molecular weight of chondroitin sulfate ranges from 5,000 to 50,000 daltons and contains about 15 to 150 basic units of D-galactosamine and D-glucuronic acid. It is represented by the following structural formula:
Chondroitin sulfate A R = SO3H R1 = H
Chondroitin sulfate C R = H R1 = SO3H
Chondroitin sulfate C, primarily found in fish and shark cartilage, but also in humans, is also made up of linear repeating units of D-galactosamine and D-glucuronic acid. The amino group of D-galactosamine is acetylated to give N-acetyl-galactosamine, and, in the case of chondroitin sulfate C, the sulfate group is esterified to the 6-position in N-acetyl-galactosamine. Chondroitin sulfate C is sometimes called chondroitin 6-sulfate. Chondroitin sulfate B is also known as dermatan sulfate. It is abundant in skin and is also found in heart valves, tendons and arterial walls. Dermatan sulfate is made up of linear repeating units containing D-galactosamine and either L-iduronic acid or D-glucuronic acid. Its molecular weight ranges from 15,000 to 40,000 daltons.
The source of chondroitin sulfate used in nutritional supplements includes the cartilaginous rings of bovine trachea and pork byproducts (ears and snout). Shark cartilage and whale septum cartilage have also been used to obtain chondroitin sulfate. Chondroitin sulfate supplements are usually isomeric mixtures of chondroitin sulfate A(chondroitin 4-sulfate) and chondroitin sulfate C(chondroitin 6-sulfate).
ACTIONS AND PHARMACOLOGY
ACTIONS
The action of orally administered chondroitin sulfate has yet to be clarified. Possible actions include promotion and maintenance of the structure and function of cartilage (referred to as chondroprotection), pain relief of osteoarthritic joints and anti-inflammatory activity.
MECHANISM OF ACTION
Until the specific actions of supplemental chondroitin sulfate are determined, the mechanism of action is a matter of speculation. However, much is known about the biochemistry and physiology of chondroitin sulfate and similar molecules. Glycoproteins known as proteoglycans form the ground substance in the extracellular matrix of connective tissue. Proteoglycans are polyanionic substances of high molecular weight and contain heteropolysaccharide-side-chains covalently linked to a polypeptide-chain backbone. The polysaccharides, which include chondroitin sulfate and hyaluronic acid, make up as much as 95% of the proteoglycan structure.
The polysaccharides in proteoglycans are called glycosaminoglycans or GAGs. Chondroitin sulfate and hyaluronic acid are vital for the structure and function of articular cartilage. Chondroitin sulfate and hyaluronic acid are fundamental components of aggrecan found in articular cartilage. Aggrecan confers upon articular cartilage shock-absorbing properties. It does this by providing cartilage with a swelling pressure that is restrained by the tensile force of collagen fibers. This balance confers upon articular cartilage the deformable resilience vital to its function. Hyaluronic acid, which is also found in synovial fluid, has lubricating properties for the joint.
In the progression of degenerative joint disease or osteoarthritis, aggrecan synthesis is decreased, leading to the loss of cartilage resiliency and the pain and other symptoms that accompany osteoarthritis.
Intra-articular injections of hyaluronic acid, an FDA-approved drug, can relieve joint pain and improve mobility. This type of therapy is called viscotherapy and is believed to act by improving joint lubrication. If chondroitin sulfate were delivered into joints, some similar effects would be expected. Animal studies have shown that parenterally administered chondroitin sulfate does get into cartilage tissue as does orally administered chondroitin sulfate. There is some human data suggesting orally administered chondroitin sulfate, particularly low-molecular-weight chondroitin sulfate, is also delivered to articular tissue. There is some indication that orally administered chondroitin sulfate leads to increases in hyaluronic acid and viscosity of synovial fluid, as well as decreases in collagenase in synovial fluid. That is, glucosamine delivered into joints may inhibit enzymes involved in cartilage degradation and enhance the production of hyaluronic acid.
PHARMACOKINETICS
Earlier studies using high-molecular-weight chondroitin sulfate, concluded that there was no significant absorption of this high-molecular-weight version of chondroitin sulfate. More recent studies demonstrate that there is probably significant absorption of low-molecular-weight chondroitin sulfate. Absorption appears to occur from the stomach and small intestine. There is also an indication that some chondroitin sulfate, after absorption, does enter the joint space. Studies of the pharmacokinetics of orally administered chondroitin sulfate are ongoing.
It is of interest to note that a molecule similar in many resects to chondroitin sulfate, pertosan polysulfate, FDA-approved for the treatment of interstitial cystitis, is given orally and is absorbed to some extent.
INDICATIONS AND USAGE
Low-molecular-weight oral chondroitin sulfate may be indicated for the treatment and prevention of osteoarthritis, either by itself or in combination with a glucosamine supplement (see Glucosamine). There is a suggestion that chondroitin sulfate may be helpful in atherosclerosis, but more research is needed to determine if this is the case.
RESEARCH SUMMARY
Two recent meta-analyses indicate that chondroitin sulfate may be useful in the treatment of osteoarthritis. One of these meta-analyses included all double-blind, placebo-controlled trials that lasted four weeks or longer. This meta-analysis also included trials that studied the effects of glucosamine (see Glucosamine) on osteoarthritis. In all, there were 13 of these studies (six involving glucosamine and seven involving chondroitin sulfate).
All 13 studies found positive results in hip or knee osteoarthritis. The authors of the meta-analysis judged a trial positive if there was 25% or more improvement in the treatment group compared with placebo. The Levesque Index and global pain scores were used to assess improvement. Very significant improvement was associated with both glucosamine (39.5%) and chondroitin (40.2%), compared with placebo.
In another recent meta-analysis of chondroitin sulfate, this one examining four randomized double-blind, placebo- or NSAID-controlled studies of 227 patients, chondroitin sulfate supplemented subjects showed at least 50% improvement, compared with controls. Various studies have reported significant reduction in NSAID use among osteoarthritis subjects supplemented with chondroitin sulfate.
There is also radiological evidence of chondroitin’s possible efficacy in osteoarthritis. Knee joint space decreased significantly in placebo subjects but remained unchanged in those receiving chondroitin sulfate for a year. And, in another study, those receiving chondroitin sulfate showed significantly fewer instances of erosive osteoarthritis (compared with placebo controls) on hand radiographs over a three-year period.
A significant synergistic effect has been reported recently using combined glucosamine hydrochloride and chondroitin sulfate in an experimental study. The combination was more effective than either substance alone in inhibiting progression of degenerative cartilage lesions. Longer term clinical studies are needed to confirm or refute this synergy effect. A large multi-center study directed by the National Institutes of Health is now underway and may shed further light on this issue.
It is believed that chondroitin sulfate’s possible efficacy in osteoarthritis derives from the fact that it is one of the two most abundant glycosaminoglycans (GAGs) in articular cartilage. Supplementation with this GAG seems, in part at least, to confer chondroprotection through its inhibitory action on some of the enzymes that damage cartilage. Further, by inhibiting other enzymes that can block transport of nutrients that nourish cartilage, this GAG may promote cartilage replacement.
It has been known for some time that injections of hyaluronic acid into arthritic joints can bring significant pain relief and enhanced mobility. Thus it is logical to assume that chondroitin sulfate, if it can reach the joints, may have similar effects since this substance has the ability to bind to receptor sites on synovial cell surfaces and thus induce production of hyaluronic acid, crucial to joint mobility.
The question for some time was whether a large molecule like chondroitin sulfate could achieve this penetration. Recent studies demonstrate that a low-molecular-weight version of oral chondroitin sulfate, of the sort used in all of the U.S. clinical trials, is absorbed.
Some years ago, chondroitin sulfate was investigated for its possible use in atherosclerosis. There was some evidence that it could favorably lower lipid levels and protect against blood clotting. Atheromatous aortic lesions were prevented in animals on high-cholesterol diets.
In a clinical trial, 60 patients suffering from coronary artery disease received 2 grams of oral chondroitin sulfate daily for 900 days. During that period, 16 of 60 unsupplemented control patients suffered acute coronary incidents. Only one of the chondroitin sulfate-treated subjects had an acute coronary incident. The same research group later followed up with similarly positive results.
More research is needed before any conclusions can be drawn with respect to a possible role for chondroitin sulfate in the treatment or prevention of atherosclerosis.
CONTRAINDICATIONS, PRECAUTIONS, ADVERSE REACTIONS
CONTRAINDICATIONS
None known.
PRECAUTIONS
Because of insufficient safety data, children, pregnant women and nursing mothers should avoid using chondroitin sulfate. Because of the theoretical possibility that chondroitin sulfate may have antithrombotic activity, those taking warfarin and those with hemophilia should exercise caution in its use. Those who need to restrict their salt intake should , if they use chondroitin sulfate, use salt-free preparations.
ADVERSE REACTIONS
Side effects that have been reported are mostly of the mild gastrointestinal variety, such as epigastric distress, nausea and diarrhea. No sulfa-allergic reactions or other allergic reactions have yet been reported.
INTERACTIONS
There are no known drug, nutrient, food or herb interactions. Chitosan (see Chitosan) may form complexes with chondroitin sulfate decreasing its absorption. Therefore, chondroitin sulfate should not be used concomitantly with chitosan.
OVERDOSAGE
Overdosage of chondroitin has not been reported in the literature.
DOSAGE AND ADMINISTRATION
Low-molecular-weight chondroitin sulfate is available as a stand-alone supplement or in combination with glucosamine (see Glucosamine). The usual dose used by those with osteoarthritis is 1,200 milligrams daily in divided doses.
It usually takes several weeks of supplementation before effects, if any, are experienced.
Chondroitin sulfate in combination with hyaluronic acid is available as an FDA-approved drug. It is used as a viscoelastic agent in cataract surgery. Hyaluronic acid itself is FDA approved for the treatment of osteoarthritis. The two forms presently available, Hylan G-F 20 (Synvisc, Wyeth-Ayerst) and sodium hyaluronate (Hyalgan, Sanofi/Orthologic), are given by intra-articular injection.
HOW SUPPLIED
Capsules — 250 mg, 400 mg, 500 mg
Powder
Tablets — 250 mg, 400 mg, 600 mg
LITERATURE
Baici A, Horler D, Moser B, et al. Analysis of glycosaminoglycans in human serum after oral administration of chondroitin sulfate. Rheum Int. 1992; 12:81-88.
Bartolucci C, Cellai L, Cordani D, et al. Chondroprotective action of chondroitin sulfate. Competitive action of chondroitin sulfate on the digestion of hyaluronan by bovine testicular hyaluronidase. Int J Tiss Res. 1991; 13:311-317.
Bourgeois P, Chales G, Dehais J, et al. Efficacy and tolerability of chondroitin sulfate 1,200 mg/day vs. chondroitin 400 mg/day vs placebo. Osteoarthritis Cartilage. 1998; 6 SupplA:25-30.
Busci L, Poor G. Efficacy and tolerability of oral chondroitin sulfate as a symptomatic slow-acting drug. for osteoarthritis (SYSADOA) in the treatment of knee osteoarthrosis. Osteoarthritis Cartilage. 1998; 6 SupplA:31-36.
Conte A, Volpi N, Palmiera L, et al. Biochemical and pharmacokinetic aspects of oral treatment with chondroitin sulfate. Drug Res. 1995; 45:918-925.
Deal CL, Moskowitz RW. Nutraceuticals as therapeutic agents in osteoarthritis. The role of glucosamine, chondroitin sulfate, and collagen hydrolysate. Rheum Dis Clin North Am. 1999; 25:379-395.
Leffler CT, Phillipi AF, Leffler SG, et al. Glucosamine, chondroitin, and manganese ascorbate for degenerative joint disease of the knee or low back: a randomized, double-blind, placebo-controlled pilot study. Mil Med. 1999; 164:85-91.
McAlindon TE, LaValley MP, Gulin JP, Felson DT. Glucosamine and chondroitin for treatment of osteoarthritis. A systematic quality assessment and meta-analysis. JAMA. 2000; 283:1469-1475.
Morrison LM, Enrick L. Coronary heart disease: reduction of death rate by chondroitin sulfate A. Angiology. 1973; 24:269-287.
Morrison LM, Bajwa GS, Alfin-Slater RB, Ershoff BH. Prevention of vascular lesions by chondroitin sulfate A in the coronary artery and aorta of rats induced by a hypervitaminosis D, cholesterol-containing diet. Atherosclerosis. 1972; 16:105-118
Pipitone VR. Chondroprotection with chondroitin sulfate. Drugs Exp Clin Res. 1991; 17:3-7.
Ronca F, Palmieri L, Panicucci P. Ronca G. Anti-inflammatory activity of chondroitin sulfate. Osteoarthritis Cartilage. 1998; 6 SupplA:14-21.
Towheed TE, Anastassiades TP. Glucosamine and chondroitin for treating symptoms of osteoarthritis. Evidence is widely touted but incomplete. JAMA. 2000; 283:1483-1484.
Uebelhart D, Thonar EJ, Delmas PD, et al. Effects of oral chondroitin sulfate on the progression of knee osteoarthritis: a pilot study. Osteoarthritis Cartilage. 1998; 6 SupplA:39-46.
Yamanashi S, Toyoda H, Furuya N, et al. Metabolic study on chondroitin sulfate in rabbits. Yakugaku Zasshi. 1991; 111:73-76.
Vitamin C
TRADE NAMES
Asco-Caps-1000 (The Key Company), Asco-Caps-500 (The Key Company), Ester-C (Swanson Health Products), C-Time (Time-Cap Labs), C-Tym (Economed Pharmaceuticals), Fruit C (Freeda Vitamins), Sunkist Vitamin C (Novartis Consumer Health), Cecon (Abbott Pharmaceutical), Vicks Vitamin C (Procter & Gamble), Dull-C (Freeda Vitamins), Mega-C (Merit Pharmaceuticals), C-Max (Bio-Tech Pharmacal), Cemill (Miller Pharmacal), Cevi-Bid (Lee Pharmaceuticals), Honey C Chews (Nature’s Life), Halls Defense (Warner Lambert).
DESCRIPTION
VITAMIN C
The term vitamin C applies to substances that possess antiscorbutic activity and includes two compounds and their salts: L-ascorbic acid, commonly called ascorbic acid, and L-dehydroascorbic acid. Ascorbic acid is the major dietary form of vitamin C. The terms vitamin C, ascorbic acid and ascorbate are commonly used interchangeably.
Vitamin C is a hexose derivative, similar in structure to the six-carbon sugar glucose. It is an essential nutrient for humans, and, as pointed out by Linus Pauling in 1970, “differs from other nutrients in that it is required in the diet by only a few species of animals—man, other primates, the guinea pig, an Indian fruit-eating bat, and the red-vented barbul and some related species of Passeriform birds.” It is also an essential nutrient for Coho salmon, rainbow trout, carp and some insects. Most other animals, all higher plant species and probably all algal classes can synthesize vitamin C from glucose or other sugars. Molecules similar to ascorbic acid are made by some fungi but not by bacteria. All vitamin C requiring animals lack the enzyme L-gulano-gamma-lactone oxidase, the final step in the synthesis of ascorbic acid from glucose.
The major deficiency syndrome of vitamin C is scurvy. Symptoms of scurvy include inflamed and bleeding gums, petechiae, ecchymosis, follicular hyperkeratosis, coiled hairs, perifollicular hemorrhages, impaired wound healing, dry eyes and mouth (Sjögren’s syndrome), arthralgia, joint effusions, muscle weakness, myalgia, fatigue, depression, frequent infections, anemia, anorexia, diarrhea, and pulmonary and kidney problems that can lead to coma and death. All systems of the body are affected by scurvy.
The antiscorbutic factor was isolated from the ox adrenal cortex in 1928 by the Hungarian biochemist Albert Szent-Györgyi and his colleagues. In 1932, the American biochemist Glen King and his colleagues isolated this factor from lemon juice. Structural studies revealed this factor to be a sugar acid and, before it was named ascorbic acid, it was called hexuronic acid and godnose.
Many of the symptoms of scurvy, particularly those having to do with connective tissue, can be explained by the known biochemical roles of vitamin C, particularly its role as a cofactor for prolyl and lysyl hydroxylase, enzymes important in the formation of collagen. Collagen synthesized in the absence of ascorbic acid—as occurs in scurvy—cannot properly form fibers, resulting in blood-vessel fragility, among other defects. In the prolyl and lysyl hydroxylase reactions, as well as in most of the biochemical reactions ascorbic acid participates in, it acts as a reducing agent. In these reactions, the vitamin reduces ferric and cupric ions to their ferrous and cuprous states, forms which are required for the reactions to proceed.
Ascorbic acid is also involved in the biosynthesis of other connective-tissue components, including elastin, fibronectin, proteoglycans, bone matrix and elastin-associated fibrillin. It also appears to play a role in collagen gene expression and cellular procollagen secretion.
The fatigue and weakness of scurvy may be due to L-carnitine deficiency. Ascorbic acid is a cofactor for crucial reactions in the carnitine biosynthetic pathway.
Ascorbic acid is involved in modulating iron absorption, transport and storage. It aids in the intestinal absorption of iron by reducing ferric iron to ferrous iron and may stimulate ferritin synthesis to promote iron storage in cells. It is involved in the biosynthesis of corticosteroids, aldosterone, the conversion of cholesterol to bile acids and functions as a reducing agent for mixed-function oxidases.
For all of this, ascorbic acid is best known for its antioxidant properties and its possible role in the prevention of certain chronic degenerative disorders, such as coronary heart disease and cancer. In fact, ascorbic acid may be the most important water-soluble antioxidant in the body.
The daily dietary intake of vitamin C necessary to prevent scurvy is about 5 to 10 milligrams. Scurvy is rare in developed countries, since most people living in these countries typically consume much more than this amount.
About 90% of vitamin C in the average diet comes from fruits and vegetables. Peppers—sweet green and red peppers and hot red and green chili peppers—are especially rich in vitamin C. Other good sources include citrus fruits and juices, brussels sprouts, cauliflower, cabbage, kale, collards, mustard greens, broccoli, spinach and strawberries. Nuts and grains contain very little vitamin C. Cooking destroys vitamin C activity.
About 5% to 10% of the total vitamin C content of fresh fruits and vegetables is comprised of dehydroascorbic acid. In the case of processed foods, dehydroascorbic acid makes up about 30% of the vitamin C content. D-ascorbic acid (erythorbic acid or isoascorbic acid), the epimer of L-ascorbic acid, is frequently added to food as an antioxidant preservative. Erythorbic acid has very low vitamin C activity.
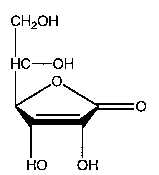
In addition to being known as ascorbic acid and L-ascorbic acid, vitamin C is also known as 2, 3-didehydro-L-threo-hexano-1, 4-lactone, 3-oxo-L-gulofuranolactone, L-threo- hex-2-enonic acid gamma-lactone, L-3-keto-threo-hexuronic acid lactone, L-xylo-ascorbic acid and antiscorbutic vitamin. It is abbreviated AA. Ascorbic acid is a crystalline, water-soluble substance with a pleasant (to some), sharp acidic taste. Its molecular weight is 176.13 daltons, and its molecular formula is C6H8O6. The structural formula of vitamin C is represented as follows:
Vitamin C
The other form of vitamin C is the oxidation product of L-ascorbic acid, L-dehydroascorbic acid or DHA.
VITAMIN C WITH BIOFLAVONOIDS
Vitamin C with bioflavonoids are mixtures of vitamin C, either as ascorbic acid or as an ascorbate, with flavonoids. Typically, the flavonoids are citrus flavonoids and are derived from lemons, oranges and grapefruits. It is believed that flavonoids work synergistically with vitamin C. This belief originates from the work and writings of the Hungarian biochemist Albert Szent-Gyögyi, the co-discoverer of ascorbic acid. Szent-Gyögyi also isolated substances from citrus fruits and Hungarian paprika which he called vitamin P. Vitamin P is now referred to as bioflavonoids or flavonoids. Flavonoids are not vitamins.
Szent-Gyögyi believed that bioflavonoids and vitamin C worked synergistically to maintain blood capillary health and prevent capillary fragility. There is some in vitro evidence that flavonoids and vitamin C do work synergistically. One study showed that ascorbic acid acts synergistically with the flavonoid quercetin to protect cutaneous tissue cells in culture against oxidative damage induced by glutathione deficiency. However, there is, as yet, no good evidence that vitamin C and flavonoids work synergistically in vivo. A recent study, in cell culture, suggested that flavonoids may even inhibit the uptake of vitamin C into cells.
Flavonoids have biological effects independent of any interaction with vitamin C. (See various monographs on flavonoids.) Flavonoids from grapefruit include quercetin, naringenin and kaempferol. Lemon flavonoids include hesperidin (hesperitin 7-0-beta-rutinoside) and eriocitrin (eriodictyol 7-0-beta-rutinoside). These flavonoids, along with rutin and others, may be found in vitamin C/bioflavonoid supplements. Some formulations use flavonoids from the sour orange Citrus aurantium.
EFFERVESCENT VITAMIN C
Effervescent vitamin C is comprised of L-ascorbic acid, citric acid and sodium bicarbonate. It is similar to Alka Seltzer with ascorbic acid added. When the tablet is placed in water, the citric acid reacts with sodium bicarbonate to form sodium citrate and carbon dioxide. Also, some sodium bicarbonate reacts with ascorbic acid to form some sodium ascorbate. Some find effervescent C a more tolerable supplement than ascorbic acid.
ACEROLA VITAMIN C
Acerola vitamin C is vitamin C derived from acerola fruit. Acerola is the fruit of the small tree or shrub known as Malphighia glabra L. Malphighia glabra is native to the Antilles and northern South America. Acerola is also known as Barbados cherry, Antilles cherry, West Indies cherry, Puerto Rican cherry, cereso, cereja-das-antilhas and cereja-do-para. In 1945, the Barbados cherry was analyzed by researchers at the School of Medicine, University of Puerto Rico, and was found to be very rich in vitamin C. Interestingly, the analysis was inspired by the use of the fruit for colds by the local people.
Acerola is one of the richest sources of vitamin C in the world. The vitamin C content of the fruit depends on ripeness, seasons, climates and localities. Content is highest when the fruit is still green and lowest when ripe. The vitamin C content of unripe fruits can range up to 4.7 grams per 100 grams of fruit or 4.7% and is about 2 grams per 100 grams or 2% in very ripe fruit. For comparison, the vitamin C content of a peeled orange is 0.05% or 50 milligrams per 100 grams. Acerola also contains flavonoids, other vitamins, such as thiamin, riboflavin, niacin, pantothenic acid and beta carotene, and minerals, such as magnesium and potassium.
Malphighia glabra has also shown active anti-fungal properties. Folk medicine uses of acerola include treatment of liver ailments, diarrhea, dysentery, coughs, colds and sore throats.
ROSE HIP VITAMIN C
Rose hips are the fruit of roses. The rose hip is the swollen ovary of the flower which produces seed after the petals of a blossom wither and fall. Once the petals have fallen off a rose all that remains attached to the stem is the rose hip. Rose hips are rich sources of Vitamin C. In fact, one species, Rosa rugosa Thunb, contains the highest amount of vitamin C of any organism in the world. Rosa rugosa Thunb rose hips can contain up to 7 grams of vitamin C per 100 grams of rose hips or 7%. Acerola, the next richest source of natural vitamin C produces up to 4.7% vitamin C, and, for comparison, the peeled orange contains 0.05% vitamin C.
During World War II, England, Norway and Sweden were faced with a scurvy crisis. Since the war had restricted normal shipping, the British could not obtain enough citrus fruit for vitamin C. Children began showing the symptoms of early scurvy. The British discovered rose hips to be an excellent source of vitamin C and made the fruit of the rose into teas, soups and syrups. The children received these supplements daily, and this prevented any problem with scurvy.
Rose hips are the major source of natural vitamin C. A few species are used to obtain the vitamin, including Rosa canina, Rosa mosqueta and Rosa rugosa Thunb. In addition to vitamin C, rose hips contain such carotenoids as beta-carotene, lycopene, zeaxanthin, rubixanthin, gazaniaxanthin, beta cryptoxanthin, gamma-carotene, lutein, violaxanthin, and antheraxanthin. They also contain flavonoids, catechins, polyphenols, procyanidins and pectins.
Rose hips have other applications. The oil extracted from its seeds is included in many cosmetic preparations for its high content of alpha-linolenic acid (45%-50%) and linoleic acid (40%). The fruit has been used as food, mainly for preparing jams, teas and alcoholic beverages.
REDUCED-ACIDITY VITAMIN C
Reduced-acidity vitamin C consists of a mixture of 50% ascorbic acid and 50% sodium ascorbate. Some find this form of vitamin C a more tolerable supplement than ascorbic acid. Since the first pKa of ascorbic acid is 4.2, the pH of the mixture dissolved in water would be 4.2. Reduced-acidity vitamin C is also known as buffered vitamin C.
NON-ACID VITAMIN C
Non-acid vitamin C consists of an ascorbate salt of sodium or calcium which has a neutral pH when dissolved in water. The calcium salt consists of two molecules of ascorbate and one atom of calcium. The molecular formula is C12H14CaO12. Calcium ascorbate is freely soluble in water. The sodium salt consists of one molecule of ascorbate and one atom of sodium. The molecular formula is C6H7NaO6. Some find sodium ascorbate and calcium ascorbate more acceptable forms for vitamin C supplementation.
ASCORBATE AND VITAMIN C METABOLITES
Ascorbate and vitamin C metabolites refer to marketed vitamin C supplements containing vitamin C in a salt form, typically as calcium ascorbate, and vitamin C metabolites. Vitamin C metabolites can include the aldonic acids L-threonic acid, L-xylonic acid and L-lyxonic acid. Typically, the vitamin C metabolite present in these products is L-threonic acid, also known as 2, 3, 4-trihydroxy- [threo] butanoic acid. L-threonic acid is usually also present as the calcium salt or calcium L-threonate, and the percentage of calcium L-threonate in the product is usually 1% of the amount of ascorbate. That is, a tablet supplying 500 milligrams of ascorbate would supply 5 milligrams of L-threonate.
Ascorbate and vitamin C metabolites are sometimes referred to as metabolite-supplemented ascorbate. Some in vitro studies have shown that the addition of L-threonate to ascorbate enhances the transfer efficiency of ascorbate into cells. Animal studies have reported increased absorption and higher retention of vitamin C when the animals were supplemented with ascorbate plus threonate than when supplemented with ascorbate alone. One cell-culture study showed that the addition of threonate to ascorbate enhanced the production of collagenous protein and mineralized tissue when compared with ascorbate alone. The authors concluded that this finding could have relevance with respect to wound healing and bone regeneration.
Although the in vitro and animal studies appear interesting, what is wanting are well-designed and well-executed clinical trials in humans to determine if vitamin C metabolites, such as L-threonate, positively affect vitamin C status.
ACTIONS AND PHARMACOLOGY
ACTIONS
Vitamin C has antioxidant activity. It may also have anti-atherogenic, anticarcinogenic, antihypertensive, antiviral, antihistaminic, immunomodulatory, opthalmoprotective and airway-protective actions. Vitamin C may aid in the detoxification of some heavy metals, such as lead and other toxic chemicals.
MECHANISM OF ACTION
Vitamin C is arguably the most important water-soluble biological antioxidant. It can scavenge both reactive oxygen species and reactive nitrogen species. Ascorbic acid or, more specifically, ascorbate is an excellent reducing agent, and it acts as a cofactor in various biochemical reactions to reduce the transition metals, iron and copper.
Ascorbate can be oxidized by most reactive oxygen and nitrogen species thought to play roles in tissue injury associated with various diseases. These species include superoxide, hydroxyl, peroxyl and nitroxide radicals, as well as such non-radical reactive species as singlet oxygen, peroxynitrite and hypochlorite. By virtue of this scavenging activity, ascorbate inhibits lipid peroxidation, oxidative DNA damage and oxidative protein damage.
Ascorbate is oxidized by reactive oxygen and nitrogen species to the semidehydroascorbate radical that is either reconverted to ascorbate via the enzyme NADH semidehydroascorbate reductase or is converted to dehydroascorbate
Dehydroascorbate in turn can be converted back to ascorbate via glutathione-dependent enzymes or catabolized.
Ascorbate can act as a secondary antioxidant. At least in vitro, ascorbate regenerates the major lipid antioxidant alpha-tocopherol from the alpha-tocopheroxyl radical form. Ascorbate may also participate in regenerating and sparing alpha-tocopherol in vivo, though this has not been clearly demonstrated. Vitamin C does preserve intracellular reduced glutathione concentrations.
The possible anti-atherogenic activity of vitamin C may be explained in a few ways. Oxidation of low-density lipoprotein (LDL) is thought to be a key early step in atherogenesis. Vitamin C protects against LDL peroxidation by scavenging peroxyl radicals in the aqueous phase. Vitamin C may enhance endothelial function by promoting the synthesis of nitric oxide (also known as NO and EDRF for endothelium-derived relaxing factor) or by preventing its inactivation by scavenging superoxide radicals. Superoxide reacts with nitric oxide to form peroxynitrite. High concentrations of vitamin C are required to prevent the interaction of superoxide with nitric oxide, extracellularly. Although such high plasma concentrations are feasible if vitamin C is given parenterally, they are likely not to occur with oral administration of vitamin C.
As noted above, vitamin C helps preserve intracellular reduced glutathione concentrations. This activity likely helps maintain nitric oxide levels and potentiates its vasoactive effects. Oral vitamin C can reach high enough concentrations intracellularly to scavenge superoxide radicals. Thus, intracellular sources of superoxide that impair nitric oxide may be scavenged by oral vitamin C. Recently, it has been found that ascorbic acid enhances nitric oxide synthase activity by increasing intracellular tetrahydrobiopterin.
Vitamin C may modulate prostaglandin synthesis to favor the production of eicosanoids with antithrombotic and vasodilatory activity. The possible sparing and regeneration of alpha-tocopherol by vitamin C could be yet another factor in the vitamin’s possible anti-atherogenic action.
Vitamin C’s possible anticarcinogenic effects may be accounted for, in part, by its ability to detoxify carcinogens, as well as its ability to block carcinogenic processes through its antioxidant activity. Vitamin C can prevent the formation of such carcinogens as nitrosamines in foods and in the gastrointestinal tract and can detoxify such chemical mutagens and carcinogens as anthracene, benzo[a]pyrene, organochlorine pesticides and heavy metals. High concentratins of ascorbic acid in gastric juice may reduce the risk of gastric cancer by inhibiting, as noted, the formation of carcinogenic N-nitroso compounds. Additionally, increased oxidative stress to the gastric mucosa has been reported in Helicobacter pylori-associated gastritis, a condition that predisposes to gastric cancer. There is preliminary evidence that vitamin C can inhibit growth of Helicobacter pylori.
Evidence appears to suggest that vitamin C may have cancer-preventive activity, at least for certain types of cancer. However, the role of vitamin C, if any, in the treatment of cancer remains very unclear. A recent cell-culture study of human breast carcinoma lines showed vitamin C to improve the antineoplastic activity of doxorubicin, cisplatin and paclitaxel. The mechanism of the effect may be pro-oxidant, not antioxidant, activity of the vitamin in potentiating the effects of these chemotherapeutic agents. Another study suggests that the pro-oxidant form of vitamin C may upregulate some of the enzymes involved in DNA repair. This possible activity may play some anticarcinogenic role.
Vitamin C may have anti-hypertensive activity in some. The mechanism of this possible effect is a matter of speculation. Some in vitro studies show that vitamin C increases the synthesis of the vasodilatory prostaglandin PGE1. However, this may not have relevance in the regulation of vascular tone in humans. As observed above, vitamin C may help maintain nitric oxide levels and potentiate its vasoactive effects. There is an indication that vitamin C may improve endothelial-dependent vasodilation in those with essential hypertension, as well as in those with hypercholesterolemia, and may help restore nitric oxide-mediated flow-dependent vasodilation in those with congestive heart failure.
There is some evidence that vitamin C inhibits the replication of human immunodeficiency virus 1 (HIV-1) in vitro. One study showed upregulation of the expression of glucose transporter 1 (Glut1) in HIV-infected cells Glut1 is one of the transport proteins for ascorbic acid. Increased cellular concentrations of ascorbate may be toxic to HIV-infected cells due to degradation of the viral nucleic acid by the action of the pro-oxidant form of vitamin C. The mechanism of the anti-HIV effect of the vitamin in vitro, however, is unclear, as is the relevance of this finding to HIV-positive individuals.
There is no evidence that vitamin C affects the replication of the viruses that cause the common cold in vivo. There is some evidence that vitamin C supplementation decreases the incidence, severity and duration of common cold symptoms in some. It is thought that this is due, at least in part, to antihistaminic activity of vitamin C.
The possible immunomodulatory activity of vitamin C may also be due, in part, to an antihistaminic effect of the vitamin. Vitamin C may enhance neutrophilic chemotaxis indirectly by reducing immunosuppressive effects of histamine. Some studies have shown that vitamin C, in vitro, enhances mitogen-stimulated lymphocyte proliferation, delayed-type hypersensitivity (DTH) response to skin antigens, natural killer cell activity and neutrophil chemotaxis. However, other studies have shown no effect of the vitamin on these and other indices of immune function.
Some studies suggest a protective effect of vitamin C supplementation against cataracts. Age-related lens opacities are thought to be due to oxidative stress. Ocular tissue concentrates vitamin C, and the antioxidant action of the vitamin could account for its possible effect in protection against cataracts.
Vitamin C may protect against asthma and other obstructive pulmonary diseases, as well as protect the airways against the effects of allergens, viral infections and irritants in some. Allergens, viruses and irritants, including ozone, nitrogen oxides and sulfur oxides, subject the airways to increased oxidative stress which can lead to bronchoconstriction. The possible protective action of vitamin C appears clearly due to its antioxidant properties.
The antioxidant properties of vitamin C can also account for its role in protecting against the tissue-damaging effect of some toxic chemicals and heavy metals. High serum levels of ascorbic acid have been reported to be associated with a decreased prevalence of elevated blood lead levels. The mechanism of the possible lead-lowering action of vitamin C is unclear. One study compared the chelating properties of ascorbic acid and the known lead-chelating agent EDTA and found them to have equivalent activity with respect to lead.
PHARMACOKINETICS
Absorption of vitamin C from the lumen of the small intestine depends on the amount of dietary intake. At a dietary intake of 30 milligrams daily, the vitamin is nearly completely absorbed from the lumen of the small intestine into the enterocytes. At an intake of 30 to 180 milligrams daily, about 70% to 90% is absorbed. About 50% of a single dose of 1 to 1.5 grams is absorbed. The percentage of a single dose absorbed decreases with increasing amounts. For example, only 16% of a single dose of 12 grams is absorbed. Maximum vitamin C absorption of large doses is attained by ingestion of several spaced doses throughout the day rather than by a single large dose. Further, sustained-release forms of large doses will give a higher efficiency of absorption than an equivalent dose that is not sustain-released. The type of food consumed does not appear to affect the absorption of supplemental vitamin C or vitamin C found in food.
The intestinal absorption of vitamin C from foods and from supplements, up to about 500 milligrams, occurs via a sodium-dependent active transport process. At doses higher than 500 milligrams, diffusion processes come into play. The major intestinal vitamin C transporter is SVCT1 (sodium-dependent vitamin C transporter 1). Some ascorbic acid may be oxidized to dehydroascorbic acid and transported into enterocytes via glucose transporters. Dietary dehydroascorbic acid is absorbed from the lumen of the small intestine into the enterocytes in such a manner. All dehydroascorbic acid within the enterocytes is reduced to ascorbic acid via reduced glutathione, and ascorbic acid leaves the enterocytes to enter, first, the portal and, subsequently, the systemic circulation. Ascorbic acid is distributed to the various tissues of the body.
Higher levels of ascorbic acid are found in the pituitary gland, the adrenal glands, the various white blood cells and the brain. Ascorbic acid itself cannot cross the blood-brain barrier. In order to enter the brain, ascorbic acid is first oxidized to dehydroascorbic acid or DHA. DHA is then transported across the blood-brain barrier by facilitative diffusion via glucose transporter 1 (GLUT1). DHA is next transported through GLUT1 at the surface of the blood-brain barrier endothelial cells. DHA is transported out of the endothelial cells through GLUT1. DHA in the brain is reduced to ascorbic acid. Ascorbic acid, once formed, is essentially trapped in the brain since it cannot be transported through GLUT1.
Ascorbic acid appears to be transported into intestinal cells, liver cells and kidney cells by a sodium-dependent active transport process via SVCT1 (sodium-dependent vitamin C transporter 1). The transporter SVCT2 (sodium-dependent vitamin C transporter 2) appears to aid in the transport of vitamin C into the aqueous humor of the eyes. Uptake of ascorbic acid into neutrophils appears to be by facilitative diffusion via GLUT1.
Regarding the metabolism of ascorbic acid, it is oxidized to dehydroascorbic acid which can either be reduced back to ascorbic acid or hydrolyzed to diketogulonate. Other metabolites include oxalic acid, threonic acid, L-xylose and ascorbate-2-sulfate. The principal route of excretion of ascorbic acid and its metabolites is via the kidney. In order to maintain ascorbic acid homeostasis, very little unmetabolized ascorbate is excreted with dietary intakes up to about 80 milligrams daily. Renal excretion of ascorbate increases proportionately with higher doses. As mentioned earlier, as the dose of supplemental ascorbic acid increases, the percentage of its absorption proportionately decreases. Consequently, there is significant fecal excretion of ascorbic acid with high supplemental intakes of the vitamin.
INDICATIONS AND USAGE
Vitamin C may be helpful in chronic diseases characterized by oxidative damage to biological molecules. Though vitamin C also has a pro-oxidant potential under some circumstances, fears raised in that regard in recent years appear overblown. There is currently no credible evidence for vitamin C pro-oxidant damage in humans except, possibly, in rare circumstances involving iron overload.
Vitamin C’s antioxidant activity, on the other hand, is well established, and that activity may be helpful in the prevention of some cancers and cardiovascular disease. Vitamin C may also be helpful in protecting against some of the lipid oxidation caused by smoking. Vitamin C’s demonstrated ability to reduce some forms of oxidative DNA damage and indications that it may also reduce protein oxidation under some circumstances further suggest that it may be of benefit in smokers and some with chronic stress and disease, in general.
Vitamin C may also be useful as an immune stimulator and modulator in some circumstances. Claims that it is a “cure” for common colds are unsubstantiated, although several studies have shown that vitamin C can significantly reduce the duration and severity of colds in some and reduce incidence in others. There is also preliminary evidence that vitamin C can be useful in ameliorating some other respiratory infections.
Vitamin C may help prevent cataracts.
Recently it was demonstrated that vitamin C can inhibit growth of Helicobacter pylori and may thus be protective against some ulcers and gastric carcinomas. There is also the suggestion in a recent report that low serum levels of ascorbic acid may be associated with a higher incidence of gall bladder disease in women. In another recent report, vitamin C supplementation was associated with reduced risk of reflex sympathetic dystrophy after wrist fracture. It may be of benefit in some burn victims and may be helpful, generally, in promoting wound healing and gum health. It has also shown benefit in some with asthma.
RESEARCH SUMMARY
Vitamin C’s antioxidant effects are well established. It has been reported to protect plasma lipids from oxidative damage. It also significantly protects DNA and protein from various oxidative processes, as demonstrated in numerous studies.
There is still controversy around claims that vitamin C can be a dangerous pro-oxidant in humans. These claims are now generally discounted, and the research that led to these fears has been widely challenged as being flawed in a number of respects. One researcher recently reviewed this controversy and concluded: “there is nothing in current data to worry members of the public who take ascorbate supplements.”
Other researchers have also recently reviewed this controversy, noting that in vitro observations of DNA damage arising in the presence of vitamin C and redox-active transition metal ions are unlikely to have relevance in vivo. The damaging effect demonstrated in vitro, these researchers point out, “requires the availability of free, redox-active metal ions and a low ratio of vitamin C to metal ion, conditions unlikely to occur in vivo under normal circumstances. Furthermore, it was shown recently that in biological fluids such as plasma, vitamin C acts as an antioxidant toward lipids even in the presence of free, redox-active iron … there is no convincing evidence for a pro-oxidant effect of vitamin C in humans.”
On the other hand, vitamin C’s antioxidant activity is marked and appears to play an important role in its possible cardioprotective activity. Several studies have shown that vitamin C, either alone, or in combination with other nutrients significantly inhibits LDL-cholesterol oxidation. This effect is most consistent when vitamin C is combined with vitamin E and/or beta-carotene, but it has also been observed when vitamin C is used alone. In the latter case, some hypothesize that it works by sparing or recycling vitamin E, an activity that has been observed in vitro. Results have been mixed in smokers in whom lipid oxidation is a serious problem. One of the better designed studies, utilizing a particularly sensitive measure of lipid oxidation, found that heavy smokers benefited from 2,000 milligrams of vitamin C administered for only five days, as measured by a significant reduction in a specific lipid oxidation marker, the F2 isoprostane 8-epi-PGF2-alpha.
Where there have been discrepancies in results from lipid (and other) biomarkers studies, some researchers attribute these, in part, to the failure of some investigators to differentiate between subjects whose tissues are already saturated with vitamin C at baseline and those whose tissues are not thus saturated. Even dietary, non-supplemental, vitamin C intake, they argue, can readily result in saturation sufficient to rule out further reductions in oxidative damage, no matter what supplemental dose is administered.
Vitamin C supplementation has also been shown, in some studies, to significantly reduce total serum cholesterol. Some others have not shown this benefit. And there have been several observational reports associating high plasma vitamin C concentrations with higher levels of HDL-cholesterol.
Platelet aggregation has been reduced in two studies utilizing 2,000-3,000 milligrams of vitamin C daily for one to six weeks. No effect was noted on platelets in another study using 250 milligrams of vitamin C daily for eight weeks. Leukocyte adhesion to endothelium, an activity implicated in atherogenesis, was significantly inhibited in smokers receiving 2,000 milligrams of vitamin C daily for ten days.
Several studies have shown that vitamin C has positive effects on hypertension. Here, too, there have been some conflicting results, but the preponderance of evidence suggests a positive effect. Epidemiological studies also consistently show that lower vitamin C intake is associated with hypertension. In one recent randomized, double-blind, placebo-controlled study, hypertensive patients received placebo or 500 milligrams of vitamin C daily for 30 days. Vitamin C resulted in a 13 mm Hg reduction in systolic blood pressure. Placebo had no effects.
Several other studies have shown that both oral administration (1,000-2,000 milligrams) and intra-arterial infusion with vitamin C can exert significant, positive effects on vasodilation in coronary artery disease patients. Similar benefits have been found in several other test groups, including smokers and those with both type 1 and type 2 diabetes.
Vitamin C’s potential impact on incidence of heart attack, stroke and death related to cardiovascular disease may be quite significant according to the findings of several epidemiological studies. In an analysis of findings from the First National Health and Nutrition Examination Survey, researchers found that “the relation of the standardized mortality ratio (SMR) for all causes of death to increasing vitamin C intake is strongly inverse for males and weakly inverse for females.” Among males with the highest vitamin C intake, SMRs were 0.65 for all causes, 0.78 for all cancers and 0.58 for all cardiovascular disease. Among females with the highest vitamin C intake, SMRs were 0.90 for all causes, 0.86 for all cancers and 0.75 for all cardiovascular disease. Comparisons were made relative to the U.S. white population, for which the SMR was defined as 1.00.
In a 20-year followup study of a cohort of randomly selected elderly people in Britain, Scotland and Wales, mortality from stroke was highest in those with the lowest vitamin C status, as measured by dietary intake and plasma ascorbic acid concentration. Adjustments were made for age, sex and established cardiovascular risk factors. The association noted was independent of social class and other dietary variables. No association was found in this study between vitamin C status and risk of death from coronary artery disease, but the researchers noted this may have been due to the age of their observed population. “Factors that may predict premature death from coronary heart disease may become less important when measured in a population of elderly survivors,” they noted. The subjects in this cohort were 65-74 years of age.
Recently, a five-year prospective population study of 1,605 Finnish men aged 42-60, who were free of atherosclerotic heart disease at baseline, concluded with these results: risk of myocardial infarction was considerably higher among those with the lowest baseline plasma vitamin C concentrations than among those with higher levels; 13.2% of those with the lowest levels suffered MIs versus 3.8% of those with higher levels.
What made this study particularly significant was its finding that increased risk of MI, in relation to plasma vitamin C concentrations, was confined to that group of subjects who were frankly deficient in vitamin C. In men with normal to high concentrations, there was no increased risk. This may have significance for some other studies that found no benefit from vitamin C in reducing cardiovascular disease risk.
It has been established by prior research that the Finnish population suffers high mortality from coronary heart disease and that many Finnish men have low plasma ascorbate concentrations. A reviewer of the Finnish study thus concluded that the finding in this study “that only individuals who are vitamin C-deficient are at increased risk may explain to some extent why no significant relationship was observed in many studies of relatively well-nourished populations.”
This observation might apply, some believe, to the Nurses’ Health Study and the Health Professionals’ Study, both followup investigations that showed a relationship between increased vitamin E intake and reduced coronary heart disease risk but no similar relationship with respect to vitamin C.
As the reviewer further observed: “Both of these studies involved generally health-conscious study subjects. The vast majority of antioxidant-disease studies, even controlled intervention studies, involve generally healthy, well-nourished populations, primarily because these populations are much easier to study. The Finnish study results, therefore, provide a special perspective that may help us to understand the mixed results from past studies and better plan future studies.”
Vitamin C has, experimentally, demonstrated an ability to protect against various cancers, most likely through its ability to inhibit DNA oxidation, through reactive nitrogen species scavenging and other antioxidant actions, as well as through its possible effects on the immune system, among other activities. There are numerous epidemiological and case-control studies showing a consistent relationship between higher dietary intakes of vitamin C and lower incidence of cancer, particularly colo-rectal, stomach, lung, breast, esophageal, oral cavity and larynx-pharynx cancers. In one review of 75 epidemiologic studies, 54 found significant evidence of reduced cancer risk in those with higher dietary vitamin C intake.
Several in vitro and animal studies have demonstrated benefits. Results of some animal studies suggested that vitamin C therapy could reduce the toxicity and/or increase the effectiveness of some standard cancer therapies. Currently some researchers have expressed fear that vitamin C might reduce the effectiveness of some radiation and cancer chemotherapies by reducing their toxicity in cancer cells, as well as in normal ells. This idea has neither been confirmed nor refuted in animal or human studies and requires further investigation. Meanwhile, other researchers have expressed doubts about this hypothesis. They point out, as noted above, that several experimental studies indicate that high doses of vitamin C not only protect normal cells from toxic cancer therapies but may simultaneously fight the cancer cells, as well.
Many population studies have found evidence of a vitamin C protective effect against some cancers. Some other studies, however, have been negative. One group of researchers reported a significant 29% reduction in risk of all cancer in males consuming 113 milligrams or more of vitamin C daily, compared with males consuming less than 82 milligrams daily. Another found that consumption of 300 milligrams of vitamin C daily, derived from diet and from supplementation, was associated with a 21% reduction in risk from all cancers in men compared with daily consumption of less than 49 milligrams daily.
In a review of many of the epidemiological studies, the authors noted: “Interestingly, virtually all of the studies in which vitamin C intakes were greater than 87 milligrams a day in the lowest intake group (quantile) found no or nonsignificant effects on cancer risk reduction with higher intakes of vitamin C …. More studies investigating cancer risk in persons with lower vitamin C intakes are warranted.”
Studies of those using higher dose vitamin C supplements have generally not shown protective effects against cancer, “possibly,” these reviewers observed, “because the dietary intake of vitamin C was already sufficient for tissue saturation.” Intervention trials with high dose vitamin C have also been mostly negative.
At present, it appears that vitamin C helps protect against a number of cancers, and the amounts of vitamin C needed for this protection can generally be obtained from a diet that includes several servings of fruits and vegetables daily—or from low-dose vitamin C supplementation. More research will be needed before vitamin C’s role, if any, in treating, as opposed to preventing cancer is established.
Vitamin C has shown a variety of activities in the immune system. It has been shown, in animal and in vitro studies, to favorably modulate lymphocytes and phagocytes. It can regulate natural killer cells under some circumstances and affect production of cytokines, antibodies and complement components.
Because supplemental vitamin C was not shown, in several studies, to reduce the incidence of the common cold, many concluded that it was of no use whatever in colds. That is still the impression of some physicians, but it is probably an erroneous one. First, a few studies have, in fact, shown a reduction in incidence of colds. Most studies have been done in normally nourished subjects in western countries; these have, typically, shown no effect on incidence. But in three trials of subjects under acute physical stress, vitamin C supplementation resulted in a 50% reduction in common cold incidence. And in four British trials, there was an average 30% reduction in incidence among those receiving vitamin C. Dietary vitamin C intake is known to be low in the UK.
Placebo-controlled trials have consistently found that supplemental vitamin C, in doses of 1 gram or greater daily, alleviated the duration and severity of cold symptoms. In several of these studies, the alleviation has been significant. For unexplained reason, there seems to be a greater effect in children than in adults and possibly, a greater effect in males than in females. The best results have been obtained with 2-gram (or greater) daily doses. There was a 6% median reduction in cold duration in five studies in which adults were administered 1 gram of vitamin C daily. There was a median decrease of 26% in two studies of children given 2 grams of vitamin C daily.
Vitamin C has also been found to be of benefit in patients with pneumonia and bronchitis. Incidence of pneumonia was significantly reduced in three controlled vitamin C studies, and substantial vitamin C treatment benefit was noted in elderly UK patients hospitalized with pneumonia or bronchitis.
There is evidence that supplemental vitamin C can inhibit the growth of Helicobacter pylori in both in vitro and animal studies. Thus it might have the potential to reduce the incidence of H. pylori-induced ulcers and subsequent gastric carcinoma. In vitro, high concentrations of vitamin C inhibited up to 90% of H. pylori growth. There was also significant inhibition of growth in animal experiments using oral administration of vitamin C.
High intake of vitamin C is strongly associated with reduced incidence of cataracts, according to the findings of case-control studies. In one study, intake of 300 milligrams or more per day was associated with a 70% reduction in risk. Another study found a 75% reduction in risk with daily intake of 490 milligrams or more per day, compared with intakes less than 125 milligrams per day. An intervention study using 120 milligrams of vitamin C daily produced a nonsignificant reduction in cataract risk of 22%, but a significant 36% reduction was observed in the same trial in subjects who consumed a multivitamin/mineral supplement.
Laboratory work has shown that vitamin C can slow chemical reactions that lead to cataracts by causing various lens proteins to aggregate. This has been demonstrated in animal work and in the human eye.
In a study of women who took vitamin C for at least ten years, incidence of cataract was significantly reduced compared with controls who did not take vitamin C. The vitamin C-supplemented women were only 23% as likely to develop cataracts compared with the women who did not take supplements. In women not taking supplements, mean daily dietary intake of vitamin C was 130 milligrams per day, about twice the recommended intake but still less than one-third the average of women taking supplements.
Recently, serum ascorbic acid levels were found to be inversely related to prevalence of gall bladder disease among women but not among men. Previously, it was shown that vitamin C-deficient guinea pigs have a high incidence of gallstones. Further clinical investigation is warranted.
In another recent study, this one a double-blind, placebo-controlled trial of vitamin C in patients with conservatively treated wrist fractures, treatment with 500 milligrams of vitamin C daily for 50 days significantly reduced the incidence of reflex sympathetic dystrophy (RSD). Followup continued for one year. The researchers proposed that “this simple and cheap means of prevention could also be useful in the prophylaxis of RSD after other injuries, such as trauma of the foot or ankle, talar and calcaneal fractures, or crural fractures.”
It was the use of vitamin C as an antioxidant therapy in dermal burns that led the researchers to believe that an antioxidant therapy might also be of benefit in preventing post-traumatic dystrophy (after wrist fracture). Researchers have found that vitamin C helps protect endothelial cells and reduces capillary permeability by reducing lipid peroxidation after burns. Some of these same mechanisms apparently account for reported beneficial effects of vitamin C in a variety of wounds, in addition to burns. There is some evidence that supplemental vitamin C may decrease permeability of gum surface tissue and may, by that and other mechanisms, help protect against periodontal gum disease.
Evidence that vitamin C can sometimes counteract the symptoms of asthma comes, in part, from a study showing that vitamin C (taken in a 500 milligram dose 90 minutes before exercise) reduces bronchial spasms in some asthma sufferers and from another study in which 1 gram of vitamin C daily reduced airway reactivity to various harmful inhalants in asthmatics.
CONTRAINDICATIONS, PRECAUTIONS, ADVERSE REACTIONS
CONTRAINDICATIONS
Vitamin C is contraindicated in those with known hypersensitivity to the substance or to any ingredient in a vitamin C-containing product.
Rose hip vitamin C
Rose hip vitamin C is contraindicated in those with known hypersensitivity to rose hips. There are reports of allergic reactions in those working with rose hips.
PRECAUTIONS
Although oxalic acid is formed when ascorbic acid is metabolized, this is highly unlikely to cause renal problems in healthy individuals without preexisting renal problems or who are not predisposed to increased crystal aggregation. Those with preexisting kidney stone disease or a history of renal insufficiency, defined as serum creatine greater than 2 and/or creatinine clearance less than 30, should exercise caution in the use of higher than RDA amounts of vitamin C (see Dosage and Administration).
Ascorbic acid is involved in modulating iron absorption and transport. It is highly unlikely that healthy individuals who take supplemental vitamin C will have any problem with iron overload. On the other hand, those with hemochromatosis, thalassemia, sideroblastic anemia, sickle cell anemia and erythrocyte G6PD deficiency might have such a problem if they use large amounts of vitamin C.
Pregnant women and nursing mothers should avoid using supplemental doses of vitamin C higher than RDA amounts.
ADVERSE REACTIONS
In healthy adults, oral doses up to 3 grams daily of vitamin C are unlikely to cause adverse reactions. The most common adverse reaction in those who take oral doses greater than 3 grams daily are gastrointestinal and include nausea, abdominal cramps, diarrhea and flatulent distention. These reactions are attributed to the osmotic effect of unabsorbed vitamin C passing through the intestine. Some advocates of megadose vitamin C use recommend titrating the daily dose of vitamin C to what they refer to as “bowel tolerance”, i.e., the point at which the user begins experiencing diarrhea. This is not recommended.
Rare adverse reactions have been reported in healthy individuals taking high oral doses of vitamin C. These include elevation of serum glucose in an adult male taking 4.5 grams daily, a gastrointestinal obstruction in a 66-year-old woman taking 4.5 grams daily of ascorbic acid and esophagitis in one person taking a single 500 milligram dose.
INTERACTIONS
DRUGS
Aluminum-containing antacids: The intake of large doses of vitamin C used at the same time as aluminum-containing antacids has been reported to increase urinary aluminum excretion, suggesting increased aluminum absorption from these antacids. However, this is not well documented.
Aspirin: Chronic use of high dose aspirin may lead to impaired vitamin C status.
Chemotherapeutic agents: Vitamin C may potentiate the antineoplastic activity of cisplatin, doxorubicin and paclitaxel. It may also help ameliorate the cardiotoxic effect of doxorubicin and the nephrotoxic effect of cisplatin. This is based on in vitro and animal studies. There is a concern by some researchers that supplemental doses of vitamin C may diminish the efficacy of some chemotherapeutic agents.
Estrogen: Ascorbic acid may enhance 17 beta-estradiol inhibition of oxidized LDL formation.
Vitamin C/Bioflavonoid combinations and drugs that inhibit cytochrome P-450 3A4: Preparations containing grapefruit flavonoids may interact with some drugs. Some drugs have up to a three-fold greater bioavailability when coadministered with grapefruit juice. It is thought that the grapefruit flavonoid naringenin plays some role in this effect. Naringenin and/or other substances found in grapefruit juice inhibit cytochrome P-450 3A4 (CYP 3A4). Drugs affected include the calcium channel blocker felodipine, as well as carbamazepine, cyclosporine, lovastatin, simvastatin, saquinavir and nisoldipine. Those taking these drugs need to exercise some caution in the use of any grapefruit products.
NUTRITIONAL SUPPLEMENTS
Copper: One study showed that high doses of vitamin C negatively ffected copper status in men. Other studies have not shown such effects.
Flavonoids: Vitamin C may act synergistically with various flavonoids. This is the basis of combining flavonoids with vitamin C in some supplements. However, it is not known if any synergism occurs to any extent in humans. There is a report that the vitamin acts synergistically with the flavonoid quercetin to protect cutaneous cells against oxidative damage. The study was performed with cells in culture. There are other reports, again from cell culture studies, that certain flavonoids such as quercetin and hesperetin may inhibit the uptake of vitamin C into cells.
Glutathione: Ascorbic acid may help maintain reduced glutathione levels in cells.
Iron: Vitamin C used concomitantly with nonheme iron supplements may increase the uptake of iron. This may cause problems in those with high iron stores or with propensity for iron overload, such as those with hemochromatosis, sideroblastic anemia, sickle cell anemia, thalassemia and erythrocyte G6PD deficiency.
Selenium: One animal study reported that the protective effect of selenite in tumorogenesis was nullified by vitamin C. The chemopreventive action of selenomethionine, a form of selenium derived from foods, was not affected by the vitamin. Selenite may be reduced by vitamin C to a form that is not available for uptake by tissue.
Vitamin E: Vitamin C may regenerate or spare d-alpha-tocopherol. However, this is based on in vitro and animal studies. It is not yet known if this occurs in humans and, if it does, to what extent.
LABORATORY TESTS
Bilirubin assay: High intakes of vitamin C may cause falsely elevated bilirubin values.
Creatine assay: Large intakes of vitamin C may cause falsely elevated urine and serum creatinine levels. However, this is not well documented.
Glucose assay: Large intakes of vitamin C may cause false positive glucose readings measured by copper reduction methods (e.g., Clinitest) and false negative glucose results as measured by the oxidase methods (e.g., Clinistix and Tes-Tape).
Guaiac assay for occult blood: Intakes of vitamin C greater than 1 gram daily may cause a false negative guaiac test.
OVERDOSAGE
There are no reports of vitamin C overdosage in the literature.
DOSAGE AND ADMINISTRATION
A dose of 200 milligrams daily is almost enough to maximize plasma and lymphocyte levels. Doses of vitamin C vary from those equivalent to the RDAs up to 5 to 10 grams daily and, in some, even higher. Typical doses used range from 500 milligrams to 2 grams daily. Some increase their dose to 4 to 5 grams daily when coming down with a cold. Such doses may have antihistaminic action. A dose of vitamin C of 5 grams daily for 4 weeks was found to significantly inhibit Helicobacter pylori in one report. Although a dose of 200 milligrams daily is almost enough to maximize plasma and lymphocyte levels, high doses may aid in detoxifying some carcinogens in the stomach prior to absorption of the vitamin.
Absorption of supplemental vitamin C is most efficient if spaced throughout the day or if taken in time-release form.
In the United States, the average intake of vitamin C is about 95 milligrams for women and 107 milligrams for men. Children between the ages of one to five consume about 83 milligrams daily.
The most recent (2000) dietary reference intakes (DRI) for vitamin C are as follows:
| Infants |
Adequate Intake (AI) |
| 0 — 6 months |
40 milligrams daily or 6mg/kg |
| 7 — 12 months |
50 milligrams daily or 6mg/kg |
|
Recommended Dietary Allowances |
|
| Children |
(RDA) |
| 1 — 3 years |
15 mg daily |
| 4 — 8 years |
25 mg daily |
| Boys | |
| 9 — 13 years |
45 mg daily |
| 14 — 18 years |
75 mg daily |
| Girls | |
| 9 — 13 years |
45 mg daily |
| 14 — 18 years |
65 mg daily |
| Men | |
| 19 — 30 years |
90 mg daily |
| 31 — 50 years |
90 mg daily |
| 51 — 70 years |
90 mg daily |
| 70 years and older |
90 mg daily |
| Women | |
| 19 — 30 years |
75 mg daily |
| 31 — 50 years |
75 mg daily |
| 51 — 70 years |
75 mg daily |
| 70 years and older |
75 mg daily |
| Pregnancy | |
| 14 — 18 years |
80 mg daily |
| 19 — 30 years |
85 mg daily |
| 31 — 50 years |
85 mg daily |
| Lactation | |
| 14 — 18 years |
115 mg daily |
| 19 — 30 years |
120 mg daily |
| 31 — 50 years |
120 mg daily |
| Smokers | |
| Men |
125 mg daily |
| Women |
110 mg daily |
A LOAEL (Lowest-Observed-Adverse-Effect Level) of 3 grams daily has been established for vitamin C for adults. Based on this LOAEL, a Tolerable Upper Level Intake (UL) for the vitamin has been set at 2 grams daily for men and women 19 years and older.
Ascorbate and vitamin C metabolites are available in a few forms. The basic form contains calcium ascorbate and calcium L-threonate (present at 1% of the ascorbate dose). Some formulations contain such substances as flavonoids, in addition. Intravenous forms of vitamin C are also available.
HOW SUPPLIED
Vitamin C is available in the following forms and strengths for Rx use:
Injection: 222 mg/mL, 250 mg/mL, 500 mg/mL
Vitamin C is available in the following forms and strengths for OTC use:
Capsules: 100 mg, 250 mg, 500 mg, 1000 mg
Capsules, Extended Release: 500 mg, 1000 mg
Cream: 10%
Chewable Tablets: :im60 mg, 100 mg, 200 mg, 250 mg,
500 mg, 1000 mg
Granules
Liquid: 100 mg/mL, 500 mg/5 mL
Lozenges: 25 mg
Powder
Syrup: 500 mg/5 mL
Tablets: 100 mg, 250 mg, 500 mg, 1000 mg
Tablets, Extended Release: :im500 mg, 1000 mg, 1500 mg, 2000 mg
LITERATURE
Agus DB, Gambhir SS, Pardridge WM, et al. Vitamin C crosses the blood-brain barrier in the oxidized form through the glucose transporters. J Clin Invest. 1997; 100:2842-2848.
Antunes LMG, Darin JDC, Bianchi MDLP. Protective effects of vitamin C against cisplatin-induced nephrotoxicity and lipid peroxidation in adult rats: a dose-dependent study. Pharmacol Res. 2000; 41:405-411.
Bors W, Michel C, Schikora S. Interaction of flavonoids with ascorbate and determination of their univalent redox potentials: a pulse radiolysis study. Free Rad Biol Med. 1995; 19:45-52.
Bush MJ, Verlangieri AJ. An acute study on the relative gastro-intestinal absorption of a novel form of calcium ascorbate. Res Commun Chem Pathol Pharmacol. 1987; 57:137-140.
Carr A, Frei B. Does vitamin C act as a pro-oxidant under physiological conditions? FASEBJ. 1999; 13:1007-1024.
Carr AC, Tijerina T, Frei B. Vitamin C protects against and reverses specific hypochlorous acid- and chloramine-dependent modifications of low-density lipoprotein. Biochem J. 2000; 346 Pt 2:491-496.
Cooke MS, Evans MD, Podmore ID, et al. Novel repair action of vitamin C upon in vivo oxidative DNA damage. FEBS Lett. 1998; 439:363-367.
Dietary Reference Intakes for Vitamin C, Vitamin E, Selenium, and Carotenoids. Washington, D.C.: National Academy Press; 2000.
Duffy SJ, Gokce N, Holbrook M, et al. Treatment of hypertension with ascorbic acid. Lancet. 1999; 354:2048-2049.
Enstrom JE, Kanim LE, Klein MA. Vitamin C intake and mortality among a sample of the United States population. Epidemiology. 1992; 3:194-202.
Fay MJ, Bush MJ, Verlangieri AJ. Effect of aldonic acids on the uptake of ascorbic acid by 3T3 mouse fibroblasts and human T lymphoma cells. Gen Pharmacol. 1994; 25:1465-1469.
Fay MJ, Verlangieri AJ. Stimulatory action of calcium L-threonate on ascorbic acid uptake by a human T-lymphoma cell line. Life Sci. 1991; 49:1377-1381.
Friedman PA, Zeidel ML. Victory at C. Nature Med. 1999; 5:620-621.
Gamble J, Grewal PS, Gartside IB. Vitamin C modifies the cardiovascular and microvascular responses to cigarette smoke inhalation in man. Clin Science. 2000; 98:455-460.
Halliwell B. Vitamin C: poison, prophylactic or panacea? Trends Biochem Sci. 1999; 24:255-259.
Harakeh S, Jariwalla RJ, Pauling L. Suppression of human immunodeficiency virus replication by ascorbate in chronically and acutely infected cells. Proc Natl Acad SciUSA. 1990; 87:7245-7249.
Harding JJ, Hassett PC, Rixon KC, et al. Sugars including erythronic and threonic acids in the human aqueous humor. Curr Eye Res. 1999; 19:131-136.
Hemila H, Douglas RM. Vitamin C and acute respiratory infections. Int J Tuberc Lung Dis. 1999; 3:756-761.
Hodison T, Socaciu C, Ropan I, Neamtu G. Carotenoid composition of Rosa canina fruits determined by thin-layer chromatography and high performance liquid chromatography. J Pharmaceut Biomed Anal. 1997; 16:521-528.
Hornero-Méndez D, Minguez-Mosquera MI. Carotenoid pigments in Rosa mosqueta hips, an alternative carotenoid source for foods. J Agric Food Chem. 2000; 48:825-828.
Hwang J, Peterson H, Hodis HN, et al. Ascorbic acid enhances 17 beta-estradiol-mediated inhibition of oxidized low density lipoprotein formation. Atherosclerosis. 2000; 150:275-284.
Ip C. Interaction of vitamin C and selenium supplementation in the modification of mammary carcinogenesis in rats. J Nat Cancer Inst. 1986; 77:299-303.
Jacob RA. Vitamin C In: Shils ME, Olson JA, Shike M, Ross AC, eds. Modern Nutrition in Health and Disease. 9th ed. Baltimore, MD: William and Wilkins; 1999:467-483.
Jarosz M, Dzieniszewski J, Dabrowska-Ufniarz E, et al. Effects of high dose vitamin C treatment on Helicobacter pylori ingestion and total vitamin C concentration in gastric juice. Eur J Cancer Prev. 1998; 7:449-454.
Kurbacher CM, Wagner U, Kolster B, et al. Ascorbic acid (vitamin C) improves the antineoplastic activity of doxorubicin, cisplatin, and paclitaxel in human breast carcinoma cells in vitro. Cancer Letters. 1996; 103:183-189.
Kwaselow A, Rowe M, Sears-Ewald D, Ownby D. Rose hips: a new occupational allergen. J Allergy Clin Immunol. 1990; 85:704-708.
Lykkesfeldt J, Christen S, Wallock LM, et al. Ascorbate is depleted by smoking and repleted by moderate supplementation: a study in male smokers and nonsmokers with matched dietary intakes. Am J Clin Nutr. 2000; 71:530-536.
Markham RG. Compositions and methods for administering vitamin C. United States Patent Number 4, 822, 816. April 18, 1989.
Moertel CG, Fleming TR, Creagan ET, et al. High-dose vitamin C versus placebo in the treatment of patients with advanced cancer who had no prior chemotherapy. A randomized double-blind comparison. N Engl J Med. 1985; 312:137-141.
Mowat C, Carswell A, Wirz A, McColl KEL. Omeprazole and dietary nitrate independently affect levels of vitamin C and nitrite in gastric juice. Gastroenterology. 1999; 116:813-822.
Ness AR, Chee D, Elliot P. Vitamin C and blood pressure—an overview. J Hum Hypertens. 1997; 11:343-350.
Panda K, Chattopadhyay R, Ghosh MK, et al. Vitamin C prevents cigarette smoke induced oxidative damage of proteins and increased proteolysis. Free Rad Biol Med. 1999; 27:1064-1079.
Park JB, Levine M. Intracellular accumulation of ascorbic acid is inhibited by flavonoids via blocking of dehydroascorbic acid and ascorbic acid uptakes in HL-60, U937 and Jurkat cells. J Nutr. 2000; 130:1297-1302.
Pauling L. Evolution and the need for ascorbic acid. Proc Natl Acad SciUSA. 1970; 67:1643-1648.
Pauling L. The significance of the evidence about ascorbic acid and the common cold. Proc Natl Acad SciUSA. 1971; 68:2678-2681.
Podmore ID, Griffiths HR, Herbert KE, et al. Vitamin C exhibits pro-oxidant effects. Nature. 1998; 392:559.
Raitakari OT, Adams MR, McCredie RJ, et al. Oral vitamin C and endothelial function in smokers: short-term improvement, but no sustained beneficial effect. J Amer Coll Cardiol. 2000; 35:1616-1621.
Rehman A, Collis CS, Yang M, et al. The effects of iron and vitamin C co-supplementation on oxidative damage to DNA in healthy volunteers. Biochem Biophys Res Commun. 1998; 246:293-298.
Rivas CI, Vera JC, Guaiquil VH, et al. Increased uptake and accumulation of Vitamin C in human immunodeficiency virus 1-infected hematopoietic cell lines. J Biol Chem. 1997; 272:5814-5820.
Rowe DJ, Ko S, Tom XM, et al. Enhanced production of mineralized nodules and collagenous proteins in vitro by calcium ascorbate supplemented with vitamin C metabolites. J Periodontol. 1999; 70:992-929.
Sakagami H, Satoh K, Hakeda Y, Kumegawa M. Apoptosis-inducing activity of vitamin C and vitamin K. Cell Mol Biol. 2000; 46:129-143.
Simon JA, Hudes ES. Relationship of ascorbic acid to blood lead levels. J Amer Med Assoc. 1999; 281:2298-2293.
Simon JA, Hudes ES. Serum ascorbic acid and gallbladder disease prevalence among US adults. The Third National Health and Nutrition Examination Survey (NHANES III). Arch Intern Med. 2000; 160:931-936.
Skaper SD, Fabris M, Ferrari V, et al. quercetin protects cutaneous tissue-associated cell types including sensory neurons from oxidative stress induced by glutathione depletion: cooperative effects of ascorbic acid. Free Rad Biol Med. 1997; 22:669-678.
Taddei S, Virdis A, Ghiadoni L, et al. Vitamin C improves endothelium-dependent vasodilation by restoring nitric oxide activity in essential hypertension. Circulation. 1998; 97:2222-2229.
Tsukaguchi H, Tokui T, Mackenzie B, et al. A family of mammalian Na+-dependent L-ascorbic acid transporters. Nature. 1999; 399:70-75.
Valkonen MM, Kuusi T. Vitamin C prevents the acute atherogenic effects of passive smoking. Free Rad Biol Med. 2000; 28:428-436.
Verlangieri AJ, Fay MJ, Bannon AW. Comparison of L-ascorbic acid and Ester C in the non-ascorbate synthesizing Osteogenic Disorder Shionogi (ODS) rat. Life Sci. 1991; 48:2275-2281.
WangY, Mackenzie B, Tsukaguchi H, et al. Human vitamin C (L-ascorbic acid) transporter SVCT1. Biochem Biophys Res Commun. 2000; 267:488-494.
Zhang HM, Wakisaka N, Maeda O, Yamamoto T. Vitamin C inhibits the growth of a bacterial risk factor for gastric carcinoma: Helicobacter pylori. Cancer. 1997; 80:1897-1903.
Zollinger PE, Tuinebreijer WE, Kreis RW, Breederveld RS. Effect of vitamin C on frequency of reflex sympathetic dystrophy in wrist fractures: a randomized trial. Lancet. 1999; 354:2025-2028.
Manganese
DESCRIPTION
Manganese is an essential trace mineral in animal nutrition and is believed to be an essential trace mineral in human nutrition, as well. Manganese is a metallic element with atomic number 25 and an atomic weight of 54.94 daltons. Its chemical symbol is Mn. Manganese exists in the oxidation states Mn2+ or Mn(II) and Mn3+ or Mn(III) under physiological conditions.
Dietary manganese-deficiency in animals results in a wide variety of structural and physiological defects, including growth retardation, skeletal and cartilage malformations, impaired reproductive function, congenital ataxia due to abnormal inner ear development, optic nerve abnormalities, impaired insulin metabolism and abnormal glucose tolerance, alterations in lipoprotein metabolism and an impaired oxidant defense system.
Manganese deficiency states have not been well documented in humans. There is one report of a man maintained for four months on a manganese-deficient diet and also given magnesium-containing antacids. The symptoms which occurred included a decrease in serum cholesterol, depressed growth of hair and nails, scaly dermatitis, weight loss, reddening of his black hair and beard and impaired blood clotting. He responded to a diet containing manganese. In another report, men fed a low-manganese diet manifested low serum cholesterol levels and dermatitis. Short-term manganese supplementation did not reverse these symptoms.
In still another report, young women fed a manganese-poor diet were found to have mildly abnormal glucose tolerance and increased menstrual losses of manganese, calcium, iron and total hemoglobin. Finally a child on long-term total parenteral nutrition (TPN) lacking manganese manifested bone demineralization and impaired growth that were corrected by supplementation with manganese.
Manganese is the preferred metal cofactor for glycosyltransferases. Glycosyltransferases are important in the synthesis of glycoproteins and glycosaminoglycans (GAGs or mucopolysaccharides). Glycoproteins are involved in the synthesis of myelin and the clotting factors, among other things. Manganese-containing metalloenzymes include manganese superoxide dismutase, the principal antioxidant enzyme of mitochondria, arginase, pyruvate carboxylase and glutamine synthetase.
The richest dietary sources of manganese include whole grains, nuts, leafy vegetables and teas. Manganese is concentrated in the bran of grains which is removed during processing. Mean intakes of manganese worldwide range from 0.52 to 10.8 milligrams daily.
ACTIONS AND PHARMACOLOGY
ACTIONS
Manganese may have antioxidant activity. Manganese has putative anti-osteoporotic and anti-arthritic activities.
MECHANISM OF ACTION
Manganese ions have been found to scavenge hydroxyl and superoxide radicals. The mechanism of binding of manganese ions to these reactive oxygen species is not known. Manganese is a crucial component of the metalloenzyme manganese superoxide dismutase (MnSOD). MnSOD is found in mitochondria and is the principal constituent of the mitochondrial oxidant defense system. Rats and mice fed manganese-deficient diets are found to have reduced MnSOD activity in heart muscle and nervous tissue. They also have mitochondrial abnormalities and pathological changes in these tissues. The pathological changes are thought to result from oxidative damage due to the decreased activity of MnSOD which normally would protect against this damage.
Dietary manganese deficiency results in skeletal and cartilage malformations in animals and in one human report. It is thought that this is due to decreased activity of the manganese-dependent glycosyltransferases which, among other things, are involved in the synthesis of glycosaminoglycans or GAGs. GAGs are crucial for healthy cartilage and bone. However, there is as yet only very preliminary evidence that supplemental manganese has any effect on the promotion of bone or cartilage formation in humans who are not manganese-deficient. One study reported that manganese when taken in combination with calcium, copper and zinc may improve bone mineral density in postmenopausal women with osteoporosis.
PHARMACOKINETICS
There is scant information on the pharmacokinetics of manganese in humans. The efficiency of absorption (fractional absorption) of ingested manganese appears to be low, about 5%. Absorption efficiency appears to decrease as dietary intake of manganese increases. It increases with low dietary intake of manganese. Absorption appears to occur throughout the small intestine and appears to occur by both active-transport and passive diffusion mechanisms. Manganese ions are transported via the portal circulation to the liver. In what forms manganese is transported to the liver—bound to albumin, alpha2-macroglobulin, hydrated manganese complexes, etc.—is also unclear. A fraction of manganese is taken up by hepatocytes and a fraction is transported by the systemic circulation to the various tissues of the body. Some manganese is bound to the plasma protein transferrin, but there also appear to be other carriers that transport manganese in the systemic circulation. Manganese is found principally in the mitochondria of cells. Absorbed manganese is excreted primarily via the biliary route. Very little manganese is excreted in the urine.
INDICATIONS AND USAGE
Apart from its uses in rare overt deficiency disorders, manganese might have some efficacy in osteoporosis and osteoarthritis as well as in some with premenstrual syndrome (PMS). Evidence for these benefits is preliminary.
RESEARCH SUMMARY
Manganese supplementation, in combination with calcium, zinc and copper, showed some efficacy in postmenopausal osteoporosis. Manganese ascorbate, in combination with glucosamine hydrochloride and chondroitin sulfate, was helpful in treating knee osteoarthritis pain in a recent randomized, double-blind, placebo-controlled pilot study. Followup on these studies is needed. Similarly, there is an isolated study needing followup that suggested some possible benefit from manganese in alleviating some PMS symptoms, including anxiety, depression, irritability and mood swings.
CONTRAINDICATIONS, PRECAUTIONS, ADVERSE REACTIONS
CONTRAINDICATIONS
Manganese supplements are contraindicated in those with liver failure. Some patients with end-stage liver disease have been found to accumulate manganese in their basal ganglia. It is thought that manganese may play a role in the hepatic encephalopathy in those with liver failure. Manganese is eliminated primarily through the bile, and hepatic dysfunction leads to depressed manganese excretion.
Manganese supplements are contraindicated in those hypersensitive to any component of a manganese-containing supplement.
PRECAUTIONS
Pregnant women and nursing mothers should avoid intakes of manganese above the upper limit of the estimated safe and adequate daily dietary intake (ESSADI). The ESSADI for those 11 years and older is 2.0 to 5.0 milligrams daily.
ADVERSE REACTIONS
Oral manganese supplements are generally well tolerated. Oral manganese, however, may be neurotoxic in those with liver failure. Manganese is primarily eliminated via the biliary route, and hepatic dysfunction leads to depressed manganese excretion. Manganese may accumulate in the basal ganglia of those with liver failure and may exacerbate hepatic encephalopathy and/or cause Parkinson’s disease-like symptoms.
Manganese is toxic under certain conditions. Hepatic failure was discussed above. Mine workers exposed to high concentrations of manganese dust develop what is known in the mining villages of northern Chile, where this disorder has been found, as “locura manganica” or manganese madness. In later stages of this disease, symptoms similar to those of Parkinson’s disease are observed. Levodopa is the treatment of the later stages of manganese madness.
There are a few reports of manganese intoxication occurring in those on long-term total parenteral nutrition (TPN) who developed parkinsonism which was treated with levodopa.
INTERACTIONS
DRUGS
Antacids: Magnesium-containing antacids, such as aluminum hydroxide/magnesium hydroxide, aluminum hydroxide/magnesium carbonate and aluminum hydroxide/magnesium trisilicate, may decrease the absorption of manganese if taken concomitantly.
Laxatives: Magnesium-containing laxatives may decrease the absorption of manganese if taken concomitantly.
Tetracycline: Tetracycline may reduce the absorption of manganese if taken concomitantly.
NUTRITIONAL SUPPLEMENTS
Calcium: Calcium supplements may decrease the absorption of manganese if taken concomitantly.
Iron: Non-heme iron supplements may reduce the absorption of manganese if taken concomitantly.
Magnesium: Magnesium supplements may decrease the absorption of manganese if taken concomitantly.
FOODS
Concomitant intake of manganese with foods rich in phytic acid (unleavened bread, raw beans, seeds, nuts and grains and soy isolates) or oxalic acid (spinach, sweet potatoes, rhubarb and beans) may depress the absorption of manganese.
DOSAGE AND ADMINISTRATION
There are several forms of supplementary manganese, including manganese gluconate, manganese sulfate, manganese ascorbate and manganese amino acid chelates. Manganese is available as a stand-alone supplement and also in combination products. One combination product used for bone/joint health contains chondroitin sulfate, glucosamine hydrochloride and manganese ascorbate.
Typical supplemental intake of manganese ranges from 2 to 5 milligrams daily.
The Food and Nutrition Board of the U.S. National Academy of Sciences has recommended the following estimated safe and adequate daily dietary intake (ESADDI) values for manganese:
| Age (years) |
ESADDI (milligrams) |
| 0 to 0.5 |
0.3 to 0.6 |
| 0.5 to 1 |
0.6 to 1.0 |
| 1 to 3 |
1.0 to 1.5 |
| 4 to 6 |
1.5 to 2.0 |
| 7 to 10 |
2.0 to 3.0 |
| 11 to 18 |
2.0 to 5.0 |
| Adults |
2.0 to 5.0 |
Up to 10 milligrams daily of manganese is considered safe.
LITERATURE.
Baly DL, Schneiderman JS, Garcia-Welsh AL. Effect of manganese deficiency on insulin binding, glucose transport and metabolism in rat adipocytes. J Nutr. 1990; 120:1075-1079.
Fell JME, Reynolds AP, Meadows N, et al. Manganese toxicity in children receiving long-term parenteral nutrition. Lancet. 1996; 347:1218-1221.
Gong H, Amemiya T. Optic nerve changes in manganese-deficient rats. Exp Eye Res. 1999; 68:313-320.
Hussain S, Ali SF. Manganese scavenges superoxide and hydroxyl radicals: an in vitro study in rats. Neuroscience Letters. 1999; 261:21-24.
Keen CL, Ensunsa JL, Watson MH, et al. Nutritional aspects of manganese from experimental studies. Neurotoxicol. 1999; 20:213-223.
Krieger D, Krieger S, Jansen O, et al. Manganese and chronic hepatic encephalopathy. Lancet. 1995; 346:270-274.
Nagatomo S, Umehara F, Hanada K, et al. Manganese intoxication during total parenteral nutrition: report of two cases and review of the literature. J Neurol Sci. 1999; 162:102-105.
Nielsen FH. Ultratrace minerals. In: Shils ME, Olson JA, Shike M, Ross AC, eds. Modern Nutrition in Health and Disease, 9th ed. Baltimore, MD: Williams and Wilkins; 1999:283-303.
Strause L, Saltman P, Glowacki J. The effect of deficiencies of manganese and copper on osteo-induction and on resorption of bone particles in rats. Calcif Tissue Int. 1987; 41:145-150
Strause L, Saltman P, Smith KT, et al. Spinal bone loss in postmenopausal women supplemented with calcium and trace minerals. J Nutr. 1994; 124:1060-1064.
Strause LG, Hegenauer J, Saltman P, et al. Effects of long-term dietary manganese and copper deficiency on rat skeleton. J Nutr. 1986; 116:135-141.
Molybdenum
TRADE NAMES
Moly-B (Carlson)
DESCRIPTION
Molybdenum is an essential trace mineral in animal and human nutrition. It is found in several tissues of the human body and is required for the activity of some enzymes that are involved in catabolism, including the catabolism of purines and the sulfur amino acids. Molybdenum is a transition metal with atomic number 42 and an atomic weight of 95.94 daltons. Its symbol is Mo. Compounds of molybdenum are among the scarcer constituents of the earth’s crust. In fact, molybdenum is only about three times more abundant than gold. The principal ore of molybdenum is molybdenite (molybdenum disulfide). Organic forms of molybdenum are found in living matter, from bacteria to animals, including humans.
In spite of its low abundance, molybdenum deficiency in humans is rare but it has been described. A patient on long-term total parenteral nutrition (TPN) developed a syndrome characterized by hypouricemia, hypermethioninemia, low urinary sulfate excretion, tachycardia, tachypnea and mental and visual disturbances. The syndrome worsened with the administration of L-methionine and the patient eventually became comatose. The patient improved when molybdenum, in the form of ammonium molybdate, was added to the TPN. The deleterious effects of molybdenum deficiency were primarily due to the accumulation of sulfite coming from the catabolism of L-cysteine. Sulfite is toxic to the nervous system and molybdenum is necessary for its metabolism to a nontoxic form.
Animals can be made molybdenum deficient by feeding them diets containing high amounts of tungsten or copper. Both tungsten and copper are molybdenum antagonists. Molybdenum deficiency has also been produced experimentally in goats by feeding them purified diets, very low in molybdenum. Molybdenum deficiency in animals results in retarded weight gain, decreased food consumption, impaired reproduction and a shortened life expectancy.
High intake of molybdenum is antagonistic to copper and can produce a condition in animals known as molybdenosis. Molybdenum-containing compounds, such as tetrathiomolybdate are currently in clinical trials for the treatment of metastatic cancer and Wilson disease. The use of copper antagonistic substances in these disorders, is known as copper depletion therapy.
ACTIONS AND PHARMACOLOGY
ACTIONS
Molybdenum prevents and treats molybdenum deficiency. Molybdenum has putative anticarcinogenic activity.
MECHANISM OF ACTION
The active biological form of molybdenum is known as the molybdenum cofactor or Moco. Moco is comprised of a molybdenum atom coordinated by the dithiolene moiety of a family of tricyclic pyranopterin structures, the simplest of which is known as molybdopterin. Moco is the cofactor for four human enzymes: xanthine dehydrogenase (xanthine: NAD+ oxidoreductase), xanthine oxidase (a form of xanthine dehydrogenase), sulfite oxidase (sulfite dehydrogenase; sulfite: ferricytochrome c oxidoreductase), and aldehyde oxidase (aldehyde: oxygen oxidoreductase). Xanthine dehydrogenase catalyzes the conversion of hypoxanthine to xanthine, and xanthine to uric acid. In addition to uric acid, the end product of purine catabolism, NADH is formed from NAD+ in the reaction. Xanthine oxidase also catalyzes the reactions of purine end metabolism. However, in the case of xanthine oxidase, which is formed from xanthine dehydrogenase, NAD+ is not a participant in the reaction, and a reactive oxygen species, the superoxide anion, is a product of the reaction.
Sulfite oxidase is involved in the degradative metabolism of the sulfur amino acids methionine and cysteine. Sulfite oxidase, which is located in mitochondria, converts sulfite to sulfate. Sulfite is derived from the metabolism of cysteine. It also enters the body in the form of free sulfites which are used as food additives. Aldehyde oxidase is involved in a number of reactions, including the catabolism of pyrimidines and the biotransformation of xenobiotics.
Deficiency of the molybdenum cofactor (Moco) causes a severe disease in humans that usually results in premature death in early childhood and is inherited as an autosomal recessive trait. All of the Moco-dependent enzymes—xanthine dehydrogenase, sulfite oxidase and aldehyde oxidase—are affected. Moco deficiency is rare. Additional signs of this combined enzyme deficiency, are severe neurological abnormalities, dislocated ocular lenses, mental retardation, increased urinary excretion of sulfite, thiosulfate, S-sulfocysteine, taurine, hypoxanthine and xanthine, and reduced serum and urine levels of sulfate and urate. Isolated sulfite oxidase deficiency is also known. This is a rare autosomal-recessive disorder presenting at birth with seizures, severe neurologic disease and lens subluxation.
Lin Xian is a small region in Honan Province in north China which has had one of the highest incidences of esophageal cancer in the world. It was determined that the soil in this area was markedly low in molybdenum. In order for nitrates in the soil to be reduced to nitrogenous substances necessary for plant nutrition, a molybdenum-dependent enzyme, nitrate reductase (found in nitrogen-fixing bacteria), is required. When the molybdenum level in the soil is low, instead of being converted to amines, the nitrates get converted to nitrosamines, known carcinogenic substances. By enriching the soil with molybdenum, as ammonium molybdate, those living in this region are exposed to lower amounts of nitrosamines in their diets, and the incidence of esophageal cancer may be declining.
The possible anticarcinogenic activity in the above example is due to feeding the soil to produce lower amounts of carcinogens. When the inhabitants in the region were administered molybdenum as a supplement, this action did not appear to affect the incidence of esophageal or any other type of cancer. However, molybdenum may have anticarcinogenic activity for a few hypothetical reasons. Aldehyde oxidase may play a role in the detoxification of some carcinogenic xenobiotics. This needs to be studied. Molybdenum is involved in cofactors that are required for enzyme activity by some of the inhabitants of the microflora of the large intestine. Some of these molybdenum-dependent enzymes may also be involved in detoxifying carcinogenic xenobiotics. This too needs study. Finally, it has been shown that copper depletion suppresses tumor growth in an animal model. There is some evidence that copper is an important cofactor for angiogenesis, and therefore, copper deficiency may suppress angiogenesis. Tetrathiomolybdate, a molybdenum compound which antagonizes copper, is now in clinical trial to determine if copper depletion therapy via molybdenum is a viable approach for the treatment of cancer.
PHARMACOKINETICS
Molybdenum in nutritional supplements is in the form of either sodium molybdate or ammonium molybdate. Molybdenum in food is principally in the form of the organic molybdenum cofactors. The efficiency of absorption of nutritional supplement forms of molybdenum ranges from 88% to 93%, and the efficiency of absorption of molybdenum from foods ranges from about 57% to 88%. Absorption of molybdenum occurs rapidly from the stomach as well as the small intestine. The mechanism of absorption—passive, active or both—is unclear. Following absorption, molybdenum is transported via the portal circulation to the liver and via the systemic circulation to the other tissues of the body. Molybdate is carried in the blood bound to alpha-macroglobulin and by adsorption to erythrocytes. The liver and kidney retain the highest amounts of molybdenum. Within cells, molybdenum participates in the formation of the molybdenum cofactor. Molybdenum is excreted in the urine as molybdate. Some molybdenum is excreted in the bile. Excretion, rather than absorption, is the principal homeostatic mechanism for molybdenum.
INDICATIONS AND USAGE
Molybdenum is indicated in cases of molybdenum deficiency due to prolonged use of total parenteral nutrition. Despite some epidemiological evidence showing a higher incidence of esophageal carcinoma in those who live in areas where the soil is low in molybdenum, there is as yet no indication for the use of supplemental molybdenum in the prevention of cancer. Claims that molybdenum may help prevent anemia, that it protects against dental caries and helps in cases of sexual impotence have no credible support.
RESEARCH SUMMARY
Except for evidence of supplemental molybdenum’s usefulness in some individuals made deficient due to prolonged total parenteral nutrition, research to date has revealed no further indications for the supplemental use of molybdenum.
An epidemiologic association has been made between the high incidence of esophageal cancer and the low intake of molybdenum in an area of China. In one study, supplementing some of those who live in this area with molybdenum for prolonged periods did not lower the incidence of cancer, although supplementation with beta carotene, vitamin E and selenium did reduce the incidence of some cancers in this study group.
Non-dietary forms of molybdenum, however, are being developed as experimental drugs for the treatment of cancer via their ability to deplete copper (see Actions and Pharmacology).
CONTRAINDICATIONS, PRECAUTIONS, ADVERSE REACTIONS
CONTRAINDICATIONS
Molybdenum is contraindicated in those who are hypersensitive to any component of a molybdenum-containing product.
PRECAUTIONS
Pregnant women and nursing mothers should avoid the use of supplemental molybdenum greater than U.S. RDA amounts (75 micrograms daily). Dietary intake of molybdenum in the United States ranges from about 120 to 240 micrograms daily, with an average intake of 180 micrograms daily. A supplementary intake of molybdenum of 75 micrograms daily brings the intake up to the upper limits of the estimated safe and adequate daily dietary intake for molybdenum.
Those with hyperuricemia and/or gout should exercise caution in the use of supplementation with doses of molybdenum greater than U.S. RDA amounts.
The use of molybdenum, specifically tetrathiomolybdate, for the treatment of cancer or Wilson disease is experimental.
ADVERSE REACTIONS
Doses of molybdenum of 10 to 15 milligrams daily have been associated with a gout-like syndrome and hyperuricemia. Supplementary doses of molybdenum of up to 500 micrograms are generally well tolerated. However, there is one report of a male who suffered acute toxicity with a molybdenum dose ranging from 300-800 micrograms daily for 18 days (see Overdosage).
INTERACTIONS
DRUGS
Acetaminophen: High doses of molybdate may inhibit the metabolism of acetaminophen.
NUTRITIONAL SUPPLEMENTS
Copper: High doses of molybdate may antagonize the absorption of copper. Likewise, high doses of copper may antagonize the absorption of molybdenum and overall decrease molybdenum status.
FOODS
High doses of molybdenum may antagonize absorption of copper from foods.
OVERDOSAGE
There is one report of acute clinical poisoning with molybdenum from a dietary molybdenum supplement. The subject, a male in his late thirties, consumed a cumulative dose of 13.5 milligrams of molybdenum over a period of 18 days, at an intake rate of 300-800 micrograms daily. This was followed by the development of acute psychosis (visual and auditory hallucinations), several petit mal seizures and one grand mal seizure. The subject was treated with chelation therapy and his symptoms and signs remitted after several hours. Neuropsychological tests and Spectral Emission Computer Tomography revealed frontal lobe damage of the brain. Major depression and learning disability persisted one year following the molybdenum incident. There are no other reports of overdosage. Further, other reports of those taking doses of molybdenum up to 500 micrograms daily or greater for extended periods of time, have not shown any adverse reactions.
DOSAGE AND ADMINISTRATION
Molybdenum supplements are usually available in the form of sodium molybdate and sometimes in the form of ammonium molybdate. Molybdenum is found in combination products, including multivitamin/multimineral formulas. A typical supplementary dose is 75 micrograms daily. The amounts of molybdenum on nutritional supplement labels, are expressed as elemental molybdenum.
The Food and Nutrition Board of the U.S. National Academy of Sciences has recommended the following estimated safe and adequate daily dietary intake (ESSADI) values for molybdenum:
| Category | Age (years) |
ESSADI |
| Infants | 0-0.5 |
15-30 |
| 0.5-1 |
20-40 |
|
| Children | 1 through 3 |
25-50 |
| 4 through 6 |
30-75 |
|
| 7 through 10 |
50-150 |
|
| 11 through 18 |
75-250 |
|
| Adults | 19 years and older |
75-250 |
The U.S. RDA for molybdenum, which is the value used for nutritional supplement and food labeling purposes, is 75 micrograms daily.
The richest dietary sources of molybdenum, include legumes, cereal grains, leafy vegetables, milk, beans, liver and kidney.
HOW SUPPLIED
Tablets — 150 mcg, 500 mcg
LITERATURE
Anon. Molybdenum deficiency in TPN. Nutr Rev. 1987; 45:337-341.
Barceloux DG. Molybdenum. J Toxicol Clin Toxicol. 1999; 37:231-237.
Barch DH. Esophageal cancer and microelements. J Am Coll Nutr. 1989; 8:99-107.
Beedham C. Molybdenum hydroxylases as drug-metabolizing enzymes. Drug Metab Rev. 1985; 16:119-156.
Blot WJ, Li JY, Taylor PR, et al. Nutrition intervention trials in Linxian, China: supplementation with specific vitamin/mineral combinations, cancer incidence, and disease-specific mortality in the general population. J Natl Cancer Inst. 1993; 85:1483-1492.
Boles JW, Klaassen CD. Effects of molybdate and pentachlorophenol on the sulfation of acetaminophen. Toxicology. 2000; 146:23-35.
Brewer GJ, Dick RD, Grover DK, et al. Treatment of metastatic cancer with tetrathiomolybdate, an anticopper, antiangiogenic agent: Phase I study. Clin Cancer Res. 2000; 6:1-10.
Brewer GJ, Johnson V, Dick RD, et al. Treatment of Wilson disease with ammonium tetrathiomolybdate. II. Initial therapy in 33 neurologically affected patients and follow-up with zinc therapy. Arch Neurol. 1996; 53:1017-1025.
Edwards MC, Johnson JL, Marriage B, et al. Isolated sulfite oxidase deficiency: review of two cases in one family. Ophthalmology. 1999; 106:1957-1961.
Hille R. Molybdenum enzymes. Essays Biochem. 1999; 34:125-137.
Johnson JL, Cohen HJ, Rajagopalan KV. Molecular basis of the biological function of molybdenum. J Biol Chem. 1974; 249:5046-5055.
Johnson JL, Waud WR, Rajagopalan KV, et al. Inborn errors of molybdenum metabolism: Combined deficiencies of sulfite oxidase and xanthine dehydrogenase in a patient lacking the molybdenum cofactor. Proc Natl Acad Sci USA. 1980; 77:3715-3719.
Luo XM, Wei HJ, Hu GG, et al. Molybdenum and esophageal cancer in China. Federation Proceedings. 1981; 46:928(Abstract#3962).
Mendel RR. The role of the molybdenum cofactor in humans. Biofactors. 2000; 11:147-148.
Momcilovic B. A case of acute human molybdenum toxicity from a dietary molybdenum supplement—a new member of the “Lucor metallicum” family. Arh Hig Rada Toksikol. 1999; 50:289-297.
Nielsen FH. Ultratrace minerals. In: Shils ME, Olson JA, Shike M, Ross AC, eds. Modern Nutrition in Health and Disease. 9th ed. Baltimore, MD: Williams and Wilkins; 1999:283-303.
Rajagopalan KV. Molybdenum—an essential trace element. Nutr Rev. 1987; 45:321-328.
Recommended Dietary Allowances. 10th Edition. Washington, DC: National Academy Press; 1989.
Thompson KH, Scott KC, Turnlund JR. Molybdenum metabolism in men with increasing molybdenum intakes: changes in kinetic parameters. J Appl Physiol. 1996; 81:1404-1409.
Turnland JR, Keyes WR, Peiffer GL. Molybdenum absorption, excretion, and retention studied with stable isotopes in young men at five intakes of dietary molybdenum. Am J Clin Nutr. 1995; 62:790-796.
Vyskocil A, Viau C. Assessment of molybdenum toxicity in humans. J Appl Toxicol. 1999; 19:185-192.
Wuebbens MW, Liu MTW, Rajagopalan KV, Schlindelin H. Insights into molybdenum cofactor deficiency provided by the crystal structure of the molybdenum cofactor biosynthesis protein MoaC. Strucutre. 2000; 8:709-718.